
Concept explainers
(a)
Interpretation:
The hybridization and approximate bond angle for each carbon atoms in
Concept Introduction:
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Geometry of different types of molecule with respect to the hybridizations are mentioned are mentioned below,
Bond angle is the angle between two bonds of a molecule and it is determined based on the electron-domain geometry.
(a)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for alanine is given below:
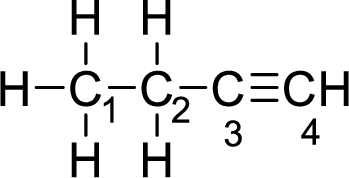 .
.
Consider the first carbon. There will be four electron regions in the molecule and hence the electron-region geometry will be tetrahedral. For a molecule having tetrahedral geometry, the hybridization will be
Consider the second carbon. There will be four electron regions in the molecule and hence the electron-region geometry will be tetrahedral. For a molecule having tetrahedral geometry, the hybridization will be
Consider the third carbon. There will be two electron regions in the molecule and hence the electron-region geometry will be linear. For a molecule having linear geometry, the hybridization will be
Consider the fourth carbon atom. There will be two electron regions in the molecule and hence the electron-region geometry will be linear. For a molecule having linear geometry, the hybridization will be
(b)
Interpretation:
The shortest carbon-to-carbon bond length in molecule has to be identified.
Concept Introduction:
Bond length is the distance between the nuclei in a bond and it is related to the sum of the covalent radii at the bonded atoms.
Higher the bond order, shorter will be the bond length.
(b)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for alanine is given below:
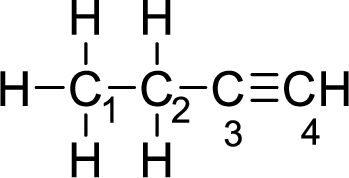 .
.
In the given molecule, the triple bond is present between
(c)
Interpretation:
The strongest carbon-to-carbon bond length in molecule has to be identified.
Concept Introduction:
Comparing triple bond, double bond and single bond, the bond with higher energy is the triple bond.
(c)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for alanine is given below:
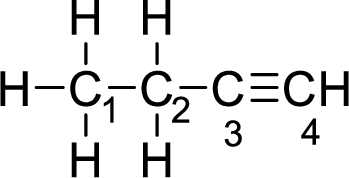 .
.
In the given molecule, the triple bond is present between
The strength of a triple bond is more than the single bond.
Thus, the strongest carbon-to-carbon bond is between
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: The Molecular Science, Hybrid Edition (with OWLv2 24-Months Printed Access Card)
- What is the hybridization of carbon in a singlet paired methylene and a triple paired methylene?arrow_forwardWhat is the Lewis Structure of CH2Se, the steric number, the electron pair geometry, the molecular geometry, and how many double bonds does it have?arrow_forwardPredict the hybridization, geometry, and bond angles for the carbon atoms in acetylene, C2H2.arrow_forward
- In C3H4 Which orbitals overlap to form the sigma bond between an outer carbon and the central carbon atom in this molecule?arrow_forwardWhat are the molecular geometry and bond angle around a sp3 carbon atom?arrow_forwardIndicate the hybridization of each of the carbon atoms in the structure below.arrow_forward
- what is the molecular geometry of cyclopropane and the hybridization ?arrow_forwardExplain the two acceptable arrangements of atoms for the molecular formula C2H6O ?arrow_forwardIdentify the type of hybridization, approximate bond angles for the N, C, and O atoms, and shortest carbon-to-oxygen bond length in alanine, an amino acid, whose Lewis structure isarrow_forward
- Write Lewis structures for the Cis -trans isomers of CH3CH = CHCl.arrow_forwardWhat is the difference between the hybridization of carbon atoms' valence orbitals in saturated and unsaturated hydrocarbons?arrow_forwardFor a given molecular formula of a hydrocarbon, such as C6H14, draw the structural formulae of its different structural isomers. For a given structural isomer, be able to draw several diagrams that all represent the same isomer that has been transformed by (a) rotation of the whole molecule and/or (b) rotation around single covalent bondsarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning


