
(a)
Interpretation:
The
Concept Introduction:
Bond angle is the angle between two bonds of a molecule and it is determined based on the electron-domain geometry.
(a)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
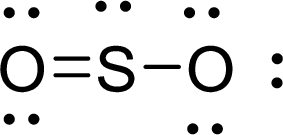
The electron-region geometry of Sulphur atom bonded to two other atoms and one lone pair of electron is triangular planar. It is a type of
(b)
Interpretation:
The
Concept Introduction:
Refer to (a).
(b)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
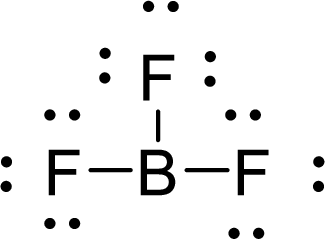 .
.
The electron-region geometry of Boron atom bonded to three other atoms is triangular planar. It is a type of
(c)
Interpretation:
The bond angles of
Concept Introduction:
Refer to (a).
(c)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
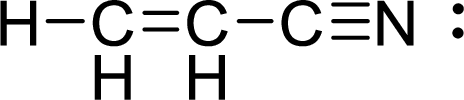 .
.
The electron-region geometry of first Carbon atom bonded to three other atoms is triangular planar. It is a type of
The electron-region geometry of third Carbon atom bonded to two other atoms is linear. It is a type of
(d)
Interpretation:
The bond angles
Concept Introduction:
Refer to (a).
(d)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
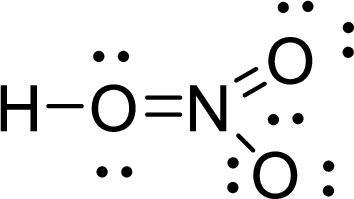 .
.
The electron-region geometry of first Oxygen atom bonded to two other atoms and two lone pair of electrons is tetrahedral. It is a type of
The electron-region geometry of nitrogen atom bonded to three other atoms is triangular planar. It is a type of
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: The Molecular Science, Hybrid Edition (with OWLv2 24-Months Printed Access Card)
- a) Methane 1CH42 and the perchlorate ion 1ClO4- 2 are both described as tetrahedral. What does this indicate about their bond angles? (b) The NH3 molecule is trigonal pyramidal, while BF3 is trigonal planar. Which of these molecules is flat?arrow_forwardWhat is the resonance form that describes the distribution of electrons in SeO2 ?arrow_forwardChemical species are said to be isoelectronic if they have the same Lewis structure (regardless of charge). Consider these ions and write a Lewis structure for a neutral molecule that is isoelectronic with them. (a) CN–, (b) NH4+ (c) CO3 2–arrow_forward
- Which has the greater bond lengths: NO2 or NO3? Explain.arrow_forwardConsider the following molecules: SiH4, PH3, H2S. In each case, a central atom is surrounded by four electron pairs. In which of these molecules would you expect the bond angle to be less than 109.5? Explain your reasoning.arrow_forwardGive two requirements that should be satisfied for a molecule to be polar. Explain why CF4 and XeF4 are nonpolar compounds (have no net dipole moments) while SF4 is polar (has a net dipole moment). Is CO2 polar? What about COS? Explain.arrow_forward
- Explain why CF4 and Xef4 are nonpolar compounds (have no net dipole moments) while SF4 is polar (has a net dipo le moment). Is CO2 polar? What about COS?arrow_forwardFormamide, HC(O)NH2, is prepared at high pressures from carbon monoxide and ammonia, and serves as an industrial solvent (the parentheses around the O indicate that it is bonded only to the carbon atom and that the carbon atom is also bonded to the H and the N atoms). Two resonance forms (one with formal charges) can be written for formamide. Write both resonance structures, and predict the bond angles about the carbon and nitrogen atoms for each resonance form. Are they the same? Describe how the experimental determination of the HNH bond angle could be used to indicate which resonance form is more important.arrow_forwardPredict die molecular structure and bond angles for each molecule or ion in Exercises 88 and 94. a. POCl3, SO42, XeO4, PO43, ClO4 b. NF3, SO32, PO33, ClO3 c.ClO2, SCl2, PCl2 d. Considering your answers to parts a, b, and c. what conclusions can you draw concerning the structures of species containing the same number of atoms and the same number of valence electrons? (O3), sulfur dioxide, and sulfur trioxide.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





