
Concept explainers
(a)
Interpretation:
The
Concept Introduction:
Bond angle is the angle between two bonds of a molecule and it is determined based on the electron-domain geometry.
(a)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
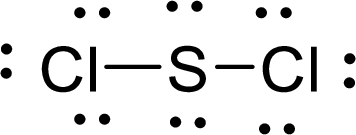 .
.
The electron-region geometry of Sulphur atom bonded to two other atoms and two lone pair of electron is tetrahedral. It is a type of
(b)
Interpretation:
The
Concept Introduction:
Refer to (a).
(b)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
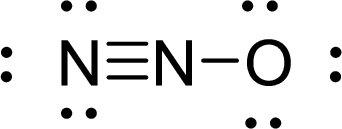 .
.
The electron-region geometry of central atom bonded to two other atoms is linear. It is a type of
(c)
Interpretation:
The bond angles of
Concept Introduction:
Refer to (a).
(c)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
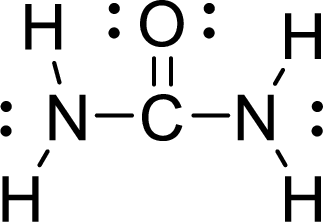 .
.
The electron-region geometry of Carbon atom bonded to three other atoms is triangular planar. It is a type of
The electron-region geometry of second Nitrogen atom bonded to three other atoms and one lone pair of electron is tetrahedral. It is a type of
(d)
Interpretation:
The bond angles of
Concept Introduction:
Refer to (a).
(d)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
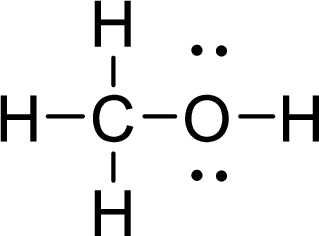 .
.
The electron-region geometry of carbon atom bonded to four other atoms is tetrahedral. It is a type of
The electron-region geometry of oxygen atom bonded to two other atoms and two lone pair of electron tetrahedral. It is a type of
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: The Molecular Science, Hybrid Edition (with OWLv2 24-Months Printed Access Card)
- For hexane, what is the bond length? Are all C—C bonds the same length?arrow_forwardYour assigneed ion is : AlCl4- please give a brief description that includes: (d) ideal bond angle, Please note that your individual assignment may be for an ion (don't forget about charges) or a neutral atom.arrow_forwardThe molecular shape of TeCl4^2¯ is __?arrow_forward
- The hypochlorite ion, ClO-, is the active ingredient inbleach. The perchlorate ion, ClO4-, is a main componentof rocket propellants. Draw Lewis structures for both ions. (a) What is the formal charge of Cl in the hypochlorite ion?(b) What is the formal charge of Cl in the perchlorate ion, assumingthe Cl—O bonds are all single bonds? (c) What is theoxidation number of Cl in the hypochlorite ion? (d) Whatis the oxidation number of Cl in the perchlorate ion, assumingthe Cl—O bonds are all single bonds? (e) In a redox reaction,which ion would you expect to be more easily reduced?arrow_forwardEach Lewis structure of benzene has three C“C double bonds. Anotherhydrocarbon containing three C“C double bonds is hexatriene, C6H8. A Lewisstructure of hexatriene is Experiments show that three of the C¬C bonds in hexatriene are shorter than theother two. Does this data suggest that hexatriene exhibits resonance structures?arrow_forwardTetrachloroethylene, C2Cl4, is used commercially as a dry-cleaning solvent. Propose a structure for tetrachloroethene based on the common bonding patterns expected in organic molecules. What kind of carbon–carbon bond ispresent?arrow_forward
- Classify the following bonds as ionic, polar covalent, or covalent, and give your reasons:(a) the CC bond in H3CCH3,arrow_forwardWrite the Lewis structure for each molecule or ion. Include resonance structures ifnecessary. Then assign the electron geometry, molecular geometry, and bond angles CN-arrow_forward(a) Describe the molecule xenon trioxide, XeO3, usingfour possible Lewis structures, one each with zero, one,two, or three Xe¬O double bonds. (b) Do any of theseresonance structures satisfy the octet rule for every atomin the molecule? (c) Do any of the four Lewis structureshave multiple resonance structures? If so, how many resonancestructures do you find? (d) Which of the Lewisstructures in part (a) yields the most favorable formalcharges for the molecule?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co




