
Interpretation:
The polar molecules has to be found from the given set of options.
Concept Introduction:
Polarity is a term that is used to explain the separation of electric charge in a molecule. A molecule is said to be polar if it contains atoms of different electronegativity bonded together. Dipole moment is the measure of polarity of the molecule. Dipole moment is the product of distance between the charges and the magnitude of electric charge, it is a vector quantity. Even though a molecule is having atoms of different electronegativity bonded together if the geometry of the molecule is symmetric, then the resultant dipole moment cancels each other and the molecule becomes non-polar.
Water is an example of polar molecule. From VSEPR theory the shape of the water is found to be bent. The direction of dipole moment is given in the Figure 1. Since the structure is not symmetrical the dipole is not cancelling each other.
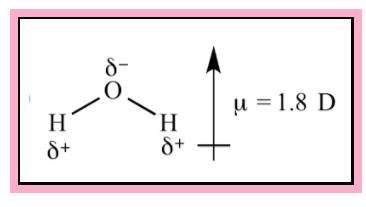
Figure 1
The linear carbon dioxide molecule is an example of non-polar molecule that is having polar bonds. Because of the symmetric structure the dipole moment cancels each other.
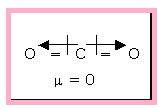
Figure 2
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
CHEMISTRY:ATOMS FIRST (LL)>CUSTOM PKG.<
- Which of the following molecules has polar covalent bonds but non-polar molecule? O2 NF3 CCl4 OF2arrow_forwardBoth of the OF2 and CS2 molecules contain polar bonds. Why one of these molecules is polar and the other is nonpolar?arrow_forwardExplain why CH3F is a polar molecule while CF4 is non-polar.arrow_forward
- Write the Lewis structure for the following molecule. State the electronic structure (shape based on electron pairs or bonds) AND the geometric structure (shape based on the atoms only). Include all valence electrons in your structure. State how many electrons are used to form covalent bonds in the molecule. Is the molecule polar? O2arrow_forwardPolarity of a molecule is important because polar molecules have different behavior and characteristics than a nonpolar molecule. O True O Falsearrow_forwardPredicting the arrangement of electron groups around the central atom of a molecule.arrow_forward
- Describe the difference between nonpolar covalent andpolar covalent bonding.arrow_forward2. If each of the bonds in these molecules are polar, which of these molecules are polar? Name the electron geometry and the molecular shape of each. polar nonpolar polar nonpolar polar nonpolar polar nonpolar polar nonpolar EG: EG: EG: EG: EG: MS: MS: MS: MS: MS:arrow_forwardDraw the Lewis structures for the following molecules. For each molecule, determine its (a) electronic geometry, (b) number of nonbonding domains on the central atom, and (c) polarity of the molecule. Remember, that molecules that have zero nonbonding domains on the central atom AND have all of the outer atoms the same are NONPOLAR. Generally, molecules with different outer atoms or with more than one nonbonding domain are POLAR. However, these may be NONPOLAR ONLY IF the dipoles cancel each other due to a symmetrical arrangement. CIF3 (a) electronic geometry: -Select-- (b) number of nonbonding domains on central atom: -Select-v (c) molecule polarity: -Select- SO2 .(a) electronic geometry: --Select- (b) number of nonbonding domains on central atom: -Select---V (c) molecule polarity: --Select--v SF5 (a) electronic geometry: [-Select- (b) number of nonbonding domains on central atom: --Select--- ♥ (c) molecule polarity: --Select-- v IFS (a) electronic geometry: -Select-- (b) number of…arrow_forward
- Peter reads in a book on Organic Chemistry that “if a molecule has resonance, it will be more stable". Will any of the ions have resonant structures? Identify those who have resonance and draw all its resonant structures. Peter also reads that the more resonant structures have a molecule, the more stable it will be. According to your criterion, Which anion will be the most stable?arrow_forwardPredicting the arrangement of electron groups around the central atom of a molecule.arrow_forward1) The question screenshot has been attached down below answer step-by-step.arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





