
Air enters the evaporator section of a window air conditioner at 100 kPa and 27°C with a volume flow rate of 6 m3/min. The refrigerant-134a at 120 kPa with a quality of 0.3 enters the evaporator at a rate of 2 kg/min and leaves as saturated vapor at the same pressure. Determine the exit temperature of the air and the rate of entropy generation for this process, assuming (a) the outer surfaces of the air conditioner are insulated and (b) heat is transferred to the evaporator of the air conditioner from the surrounding medium at 32°C at a rate of 30 kJ/min.
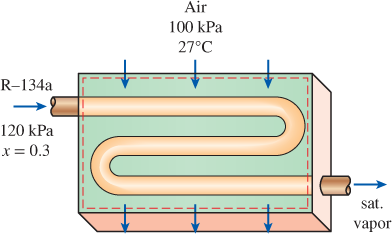
FIGURE P7–180
a)
The exit temperature of air and the entropy generated during the process.
Answer to Problem 181RP
The exit temperature of air is
The entropy generated during the process is
Explanation of Solution
Write the expression to calculate the initial enthalpy of the refrigerant.
Here, initial enthalpy is
Write the expression to calculate the initial entropy of the refrigerant.
Here, initial entropy is
Write the expression to calculate the mass flow rate of air.
Here, mass flow rate of air is
Write the expression for the mass balance of the system.
Here, mass flow rate into the control system is
Write the expression for the energy balance equation for closed system.
Here, rate of energy transfer into the control volume is
Write the expression for the rate of entropy balance for the system.
Here, rate of entropy in the system is
Conclusion:
From Table A-12, “Saturated refrigerant-134a-Pressure table”, Obtain the following properties at saturated pressure of 120 kPa
Saturated liquid enthalpy,
Evaporated enthalpy,
Saturated vapor enthalpy,
Saturated vapor entropy,
Saturated liquid entropy,
Evaporated entropy,
Substitute
Substitute
Refrigerant –134a enters and leaves at the same pressure. Hence,
From Table A-1E, “the molar mass, gas constant and critical–point properties table”, select the gas constant of air at room temperature as
Substitute
Substitute
Here, mass flow rate of refrigerant is
Substitute
Here, mass flow rate of air is
Substitute
From the Table A-2, “Ideal-gas specific heats of various common gases”, select the value of the specific heat at constant pressure value of air as
Substitute 27 C for
Hence, the exit temperature of air is
For the steady flow system, change of entropy in the system is zero.
Substitute
Substitute
Hence, the entropy generated during the process is
b)
The exit temperature of air and the entropy generated during the process.
Answer to Problem 181RP
The exit temperature of air is
The entropy generated during the process is
Explanation of Solution
Write the expression for the energy balance equation for closed system.
Here, rate of energy transfer into the control volume is
Write the expression for the rate of entropy balance for the system.
Here, rate of entropy in the system is
Conclusion:
Substitute
Here, rate of heat gain from the surrounding is
Substitute 27 C for
Hence, the exit temperature of air is
For the steady flow system, change of entropy in the system is zero.
Substitute
Substitute
Hence, the entropy generated during the process is
Want to see more full solutions like this?
Chapter 7 Solutions
Thermodynamics: An Engineering Approach
- Air at 200 kPa and 950 K enters an adiabatic nozzle at low velocity and is discharged at a pressure of 110 kPa. If the isentropic efficiency of the nozzle is 92 percent, determine the actual exit velocity of the air. Assume constant specific heats for air.arrow_forwardRefrigerant-134a enters a compressor as a saturated vapor at 160 kPa at a rate of 0.03 m3 /s and leaves at 800 kPa. The power input to the compressor is 10 kW. If the surroundings at 20°C experience an entropy increase of 0.008 kW/K, determine the exit temperature of the refrigerant.arrow_forwardDetermine the work input and entropy generation during the compression of steam from 100 kPa to 1 MPa in an adiabatic compressor if the inlet state is saturated liquid in the pump and saturated vapor in the compressor and the isentropic efficiency is 85 percent for both devices.arrow_forward
- Hot combustion gases enter the nozzle of a turbojet engine at 230 kPa, 627°C, and 60 m/s and exit at 70 kPa and 450°C. Assuming the nozzle to be adiabatic and the surroundings to be at 20°C, determine the exit velocity.arrow_forwardAir enters the compressor of a gas-turbine plant at ambient conditions of 100 kPa and 25 °C with a low velocity and exits at 1 MPa and 347 °C with a velocity of 90 m/s. The compressor is cooled at a rate of 1500 kJ/min, and the power input to the compressor is 250 kW. Determine the mass flow rate of air through the compressor. (Hint: Use Table A-17 of the Booklet for the properties of air.)arrow_forwardDetermine the work input and entropy generation during the compression of steam from 100 kPa to 1 MPa in (a) an adiabatic pump and (b) an adiabatic compressor if the inlet state is saturated liquid in the pump and saturated vapor in the compressor and the isentropic efficiency is 85 percent for both devices.arrow_forward
- Steam enters an adiabatic turbine at 8 MPa and 500°C at a rate of 18 kg/s, and exits at 0.2 MPa and 300°C. The rate of entropy generation in the turbine is (a) 0 kW/K (b) 7.2 kW/K (c) 21 kW/K (d) 15 kW/K (e) 17 kW/Karrow_forwardAir enters the evaporator section of a window air conditioner at 100 kPa and 27C with a volume flow rate of 6 m3/min. The refrigerant-134a at 120 kPa with a quality of 0.3 enters the evaporator at a rate of 2 kg/min and leaves as saturated vapor at the same pressure. Determine the exit temperature of the air and the rate of entropy generation for this process, assuming (a) the outer surfaces of the air conditioner are insulated and (b) heat is transferred to the evaporator of the air conditioner from the surrounding medium at 32C at a rate of 30 kJ/min.arrow_forwardSteam enters an adiabatic turbine steadily at 3 MPa and 400°C and leaves at 50 kPa and 100°C. If the power output of the turbine is 2 MW, determine the isentropic efficiency of the turbine.arrow_forward
- Refrigerant-134a enters an adiabatic compressor as saturated vapor at 30 psia at a rate of 20 ft3 /min and exits at 70 psia pressure. If the isentropic efficiency of the compressor is 80 percent, determine the second-law efficiency of the compressor. Assume the surroundings to be at 75°F.arrow_forwardRefrigerant-134a is to be cooled by water in a condenser. The refrigerant enters the condenser with a mass flow rate of 6 kg/min at 1 MPa and 70°C and leaves at 35°C. The cooling water enters at 300 kPa and 15°C and leaves at 25°C. Neglecting any pressure drops, determine the mass flow rate of the cooling water required.arrow_forwardSteam flows steadily through an adiabatic turbine. The inlet conditions of the steam are 4 MPa, 500°C, and 80 m/s, and the exit conditions are 30 kPa, 92 percent quality, and 50 m/s.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





