
Concept explainers
(a)
Interpretation: The effect on the rate of the given second-order reaction is to be interpreted if the concentration of chlorocyclopentane is tripled and the concentration of sodium hydroxide remains the same.
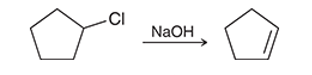
Concept introduction:
The change in reactant concentration over the course of the specified period can be viewed as the pace of a
(b)
Interpretation: The effect on the rate of the given second-order reaction is to be interpreted if the concentration of chlorocyclopentane remains the same and the concentration of sodium hydroxide is doubled.
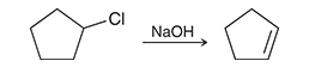
Concept introduction:
The change in reactant concentration over the course of the specified period can be viewed as the pace of a chemical reaction. According to the rate rule, the active concentration of the reactant molecules directly affects how quickly a chemical reaction proceeds. The rate constant is the name for the proportionality constant.
(c)
Interpretation: The effect on the rate of the given second-order reaction is to be interpreted if the concentration of chlorocyclopentane is doubled and the concentration of sodium hydroxide is tripled.
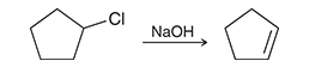
Concept introduction:
The change in reactant concentration over the course of the specified period can be viewed as the pace of a chemical reaction. According to the rate rule, the active concentration of the reactant molecules directly affects how quickly a chemical reaction proceeds. The rate constant is the name for the proportionality constant.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
ORGANIC CHEM. - ACCESS PKG+MODELING KIT
- In each of the following, which reaction mechanism assumption is apparently being violated? Explain your answers. a. A reaction takes place more rapidly when the concentration of reactants is decreased. b. A reaction takes place more rapidly when the reaction mixture is cooled. c. The reaction rate of A+BAB increases as the concentration of A is increased but does not change as the concentration of B is increased.arrow_forwardBy which of the following mechanisms does a catalyst operate? a. It decreases the activation energy barrier for a reaction. b. It serves as a reactant and is consumed. c. It increases the temperature of a reaction. d. It increases the concentration of reactants.arrow_forwardCobra venom helps the snake secure food by binding to acetylcholine receptors on the diaphragm of a bite victim, leading to the loss of function of the diaphragm muscle tissue and eventually death. In order to develop more potent antivenins, scientists have studied what happens to the toxin once it has bound the acetylcholine receptors. They have found that the toxin is released from the receptor in a process that can be described by the rate law Rate = k [acetylcholine receptortoxin complex] If the activation energy of this reaction at 37.0C is 26.2 kJ/mol and A = 0.850 s1. what is the rate of reaction if you have a 0.200M solution of receptor-toxin complex at 37.0C?arrow_forward
- How would you express the rate of the chemical reaction AB based on the concentration of Reactant A? Howwould that rate compare with the reaction rate based onthe Product B?arrow_forwardWhy awe elementary reactions involving three or more reactants very uncommon?arrow_forwardThree first-order reactions have the following activation energies: (a) Which reaction is the fastest? (b) Which reaction has the largest half-life? (c) Which reaction has the largest rate?arrow_forward
- Reaction A and reaction B have identical frequency factors, but reaction B has a higher activation energy than reaction A. Which reaction has a faster rate at room temperature? A. Reaction A B. Reaction Barrow_forwardChoose the letter of the best answer and write your answer on the space provided before the number. I. 1. Why does the rate of the reaction increase when the temperature of a reaction increase? A. activation energy is lowered B. reactant molecules collide less frequently C. reactant molecules collide more frequently and with greater energy per collision D. reactant molecules collide less frequently and with greater energy per collision 2. Which of the following lowers the activation energy for a reaction? A. adding a catalyst for the reaction B. increasing the concentrations of reactants C. raising the temperature of the reaction D. removing products the reaction as proceedarrow_forwardPLEASE COMPLETE ALL MSCQ THAN RATING HELPFULarrow_forward
- as the temp of a reaction is increased the rate of the reaction increases why? (see image)arrow_forwardWhich of the following statements regarding the rate constant in the rate law expression is not true? a. Its value increases with temperature. b. Its value is independent of initial concentration at a given temperature. c. Its units depend on the overall order of reaction. d. Its value is experimentally determined. e. The larger its value, the slower the reaction rate.arrow_forwardLabel the graph to the right with the items on the left. a. Products b. Transition state c. Activation energy d. Potential energy e. Catalyzed reaction f. Reaction progress g. Uncatalyzed reaction h. Reactants i. AH of the reactionarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





