
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.7, Problem 7.62P
Interpretation Introduction
Interpretation:
Efficient synthesis has to be proposed for the given transformation.
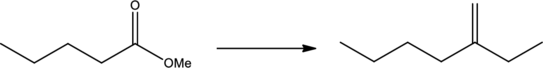
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Hello there!
I am needing some help. I need to get the product (shown on the white slip) by doing reverse synthesis. The second photo is what I had come up with, however, I need some help to make sure that I did this correctly. Can you help?
Thank you so much!
Hi, I need help to perform a retrosynthetic analysis and then to design a synthesis plan. Can you answer my question in more detail? In the RSA should all disconnections, synthons, and synthetic equivalent be shown. Can you do that step by step?
Using the retrosynthetic analysis propose a logical route for the following transformation. Draw all substrates, reagents and products
Chapter 7 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 7.3 - Propose a plausible mechanism for each of the...Ch. 7.3 - Propose a plausible mechanism for each of the...Ch. 7.3 - Propose a plausible mechanism for each of the...Ch. 7.3 - Propose a plausible mechanism for each of the...Ch. 7.3 - Propose a plausible mechanism for each of the...Ch. 7.3 - Predict the major product in each of the following...Ch. 7.3 - Predict the major product in each of the following...Ch. 7.3 - Predict the major product in each of the following...Ch. 7.3 - Predict the major product in each of the following...Ch. 7.3 - Predict the major product in each of the following...
Ch. 7.3 - Identify the reagents you would use to achieve...Ch. 7.3 - Identify the reagents you would use to achieve...Ch. 7.3 - Identify the reagents you would use to achieve...Ch. 7.3 - Identify the reagents you would use to achieve...Ch. 7.3 - Identify the reagents you would use to achieve...Ch. 7.4 - Predict the major products for each of the...Ch. 7.4 - Predict the major products for each of the...Ch. 7.4 - Predict the major products for each of the...Ch. 7.5 - Prob. 7.23PCh. 7.5 - Identify the reagents you would use to make each...Ch. 7.5 - Identify the reagents you would use to make each...Ch. 7.5 - Identify the reagents you would use to make each...Ch. 7.5 - Identify the reagents you would use to make each...Ch. 7.5 - In the space provided, draw a mechanism for each...Ch. 7.5 - In the space provided, draw a mechanism for each...Ch. 7.5 - Prob. 7.33PCh. 7.5 - Prob. 7.34PCh. 7.5 - Prob. 7.35PCh. 7.5 - Prob. 7.36PCh. 7.6 - Based on everything we have just seen, propose a...Ch. 7.6 - Prob. 7.39PCh. 7.6 - Prob. 7.40PCh. 7.6 - Prob. 7.41PCh. 7.6 - Propose a mechanism for the following reaction:Ch. 7.6 - Prob. 7.44PCh. 7.6 - Prob. 7.45PCh. 7.6 - Prob. 7.46PCh. 7.7 - Prob. 7.48PCh. 7.7 - Prob. 7.49PCh. 7.7 - Prob. 7.50PCh. 7.7 - Prob. 7.51PCh. 7.7 - Prob. 7.52PCh. 7.7 - Prob. 7.53PCh. 7.7 - Prob. 7.55PCh. 7.7 - Prob. 7.56PCh. 7.7 - Prob. 7.57PCh. 7.7 - Prob. 7.58PCh. 7.7 - Prob. 7.59PCh. 7.7 - Prob. 7.60PCh. 7.7 - Prob. 7.61PCh. 7.7 - Prob. 7.62PCh. 7.7 - Prob. 7.63PCh. 7.7 - Prob. 7.64PCh. 7.7 - Prob. 7.65P
Knowledge Booster
Similar questions
- I have proposed a suitable synthesis for the following transformation please let me know whether or not this is correct. If incorrect please let me know what I did wrong.arrow_forwardComplete the following synthesis by selecting from the list of 10 reagents below. Each reagent (or set of reagents) is labeled as a letter. In the answer box, simply place the order of reagents used as uppercase letters. For example, if your synthesis involves using reagent A followed by B,followed by C, and then D, your answer would be: ABCD.arrow_forwardSelect reagents from the above table to carry out each of the following reactions.If only a single step is required, select (none) from the drop down list.arrow_forward
- You will be performing the first step of your multistep synthesis to make trans-1,2-cyclohexanol from hexanol 1. Draw the appropriate compound next to the question mark. 2. Copy the retrosynthesis below and fill in the missing compounds. 3. The final step is to redraw the retrosynthesis as a normal reaction scheme showing each step.arrow_forwardFor the following reactions, fill in the appropriate product(s). BE SURE TO INCLUDE ANY APPROPRIATE STERECHEMISTRY. Then, circle one criterion from each of the two columns on the right.. make sure each section includes a structure, regioselectivity criterion, and stereoselectivity criterion. You may use the image below as a template for your answerarrow_forwardFirst off, I am really trying to learn this retrosynthetic-type reaction, please show and explain how you would go about completing this problem. I will check it with my work I have already done.arrow_forward
- Construct a multistep synthetic route from ethylbenzene to (2-bromoethyl)benzene by dragging the appropriate items into the bins. Note that each bin will hold only one item, and not all reagents and structures will be used.arrow_forwardProvide an efficient synthesis for each of the following transformations. Include your retrosynthetic analysis, showing products for each step of the reaction sequence. You are not required to write a mechanism for any of these.arrow_forwardIn the following reaction Series, write down the appropriate reagents that can be used where there are question marksarrow_forward
- Propose a synthesis for the transformations shown below (no mechanisms needed, just products/intermediates and reagents). You will get full credit if you figure out two out of three. Clearly indicate which two you want us to grade.arrow_forwardThe synthesis shown above can be accomplished in three steps. Select the reagents for each step from the dropdown lists below.arrow_forwardFill in the reagents necessary for this transformation.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning