
Concept explainers
a)
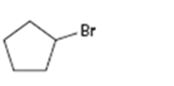
Interpretation:
The alkene that will give 1-bromocyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical
To state:
Which alkene will give 1-bromocyclopentane as product in an addition reaction.
b)
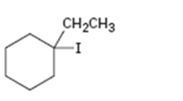
Interpretation:
The alkene that will give 1-ethyl-1-iodocyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 1-ethyl-1-iodocyclohexane as product in an addition reaction.
c)
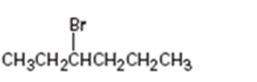
Interpretation:
The alkene that will give 3-bromohexane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 3-bromohexane as product in an addition reaction.
d)
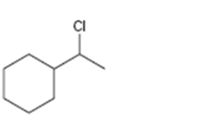
Interpretation:
The alkene that will give 1-chloroethylcyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 1-chloroethylcyclohexane as product in an addition reaction.
Trending nowThis is a popular solution!

Chapter 7 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- please answer both questionsarrow_forwardIntermidiates and reagents of the following two final products from a Acetylene Note: the final products are different reactions but use the same starting material.arrow_forwardThe pictured reaction shows an alkyl bromide being converted into an alkene. Choose all reagents that would produce the pictured alkene as the major product. A) NaOH/H2O B) H2O C) tBuOK/tBuOH D) EtONa/EtOHarrow_forward
- Could you please write the products formed in the following reaction?arrow_forward4) Draw all of the possible alkene products from the following reaction. Indicate in what proportions these alkenes would be produced relative to one another. HO H₂SO4 Refluxarrow_forwardDraw structures for the alkene (or alkenes) that gives the following reaction product.arrow_forward
- How do you synthesize a cyclohexane-1,2,3-triol from a cyclohexanearrow_forwardHow do you prepare pent-2-ol from ethyne? This is alkenes and alkynes preparationarrow_forwardThe following reaction is a/an example of H2 I H₂ ó H₂CCCC. H2 Addition Condensation Elimination Substitution Oxidation Cl H2 Cl₂and light H₂C-CH CC + HC CH3 CH3 H2 other + chlorinated productsarrow_forward
