
Concept explainers
a)
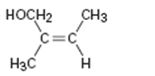
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The member that ranks higher can be determined by considering the
To assign:
The configuration for the compound given as E or Z.
b)
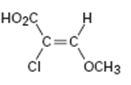
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The member that ranks higher can be determined by considering the atomic number of the first atom of the two substituents separately. The atom with highest atomic number gets a higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms away from double bond are considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms. The isomer that has the higher ranked groups on each carbon are on the same side of the double bond is said to have Z configuration. If the higher ranked groups are on opposite sides, the alkene is said to have E configuration.
To assign:
The configuration for the compound given as E or Z.
c)
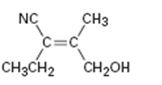
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The member that ranks higher can be determined by considering the atomic number of the first atom of the two substituents separately. The atom with highest atomic number gets a higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms away from double bond are considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms. The isomer that has the higher ranked groups on each carbon are on the same side of the double bond is said to have Z configuration. If the higher ranked groups are on opposite sides, the alkene is said to have E configuration.
To assign:
The configuration for the compound given as E or Z.
d)
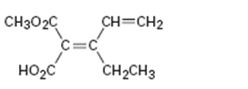
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The member that ranks higher can be determined by considering the atomic number of the first atom of the two substituents separately. The atom with highest atomic number gets a higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms away from double bond are considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms. The isomer that has the higher ranked groups on each carbon are on the same side of the double bond is said to have Z configuration. If the higher ranked groups are on opposite sides, the alkene is said to have E configuration.
To assign:
The configuration for the compound given as E or Z.
Trending nowThis is a popular solution!

Chapter 7 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Identify the relationship between the following compounds. H Br CH3 H3C CH3 HO H Hint H3C. H OH Br H enantiomers diastereomers constitutional isomer compounds with different molecular formulas O identical Can the structure be rotated about a single bond?arrow_forwardProvide a systematic name (IN SMALL LETTERS except for E/Z and R/S, NO SPACES) for each of the following compounds.arrow_forwardAssign E or Z configuration to the following alkenes:arrow_forward
- Identify the relationship between the given pairs of compounds. Choose from: A = enantiomers B = diastereomers C = constitutional isomers D = geometric isomers %3D E = conformational isomers F = two molecules of the same compound CO,H CH3 Br H H- Br H- Br H- Br CO,H CH3arrow_forwardAssign absolute configurations, R or S and E or Z to each of the compounds below.arrow_forwardA Compound may have a possible number of which is H ₂ C ₁ stereoisomers formula 2^ (n = number of stereocentres. The compound (-) menthone (below) is a Natural Product found in peppermint. Use the given formula given to determine the number of possible stereoisomers in this Compound can have. I determined with the # CH(CH3)₂ H E 19arrow_forward
- Provide a systematic name (IN SMALL LETTERS except for E/Z and R/S, NO SPACES) for each of the given compounds.arrow_forwardWhat is the degree of unsaturation of a compound with a formula of C6H8? 0 1 2 3 If a compound has a degree of unsaturation of 1, what are the possible combinations for its structure? two double bonds or one ring no double bond or two rings one double bond or one ring two double bonds or two ringsarrow_forwardIdentify the relationship between the given pairs of compounds. Choose from: A = enantiomers B = diastereomers C = constitutional isomers D = geometric isomers %3D E = conformational isomers F = two molecules of the same compound H,C CH3 Br CH3 H. Br F H,C H.arrow_forward
- 6-In each of the following pairs of compounds one is chiral and the other is achiral. Identify each compound as chiral or achiral, as appropriate. NH H OH CH, -H -NH₂ CH, OH and HO and H H CH₂ -NH₂ -NH₂ CH₂ OHarrow_forwardOne commercial synthesis of ketoprofen (the active ingredient in Actron and a score of other over-the-counter and prescription nonsteroidal anti-inflammatory drug preparations) gives the enantiomer shown in 93% enantiomeric excess. H CH3 он (a) Assign an R or S configuration to this enantiomer of ketoprofen. R S What are the percentages of R and S enantiomers in the mixture? (Enter unrounded values.) (b) R: % S: %arrow_forward6 Which of the following compounds has both optical and E-Z isomers? A CH₂CH=CHCH₂CH3 B CH₂CHBRCH=C(CH3)₂ D c CHBr=CHCHBrCH, D CH₂=CHCHBRCH3arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
