
a)
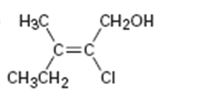
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The two groups attached to the carbons in double bond are to be ranked first. The member that ranks higher can be determined by considering the
To assign:
The configuration for the compound given as E or Z.
b)
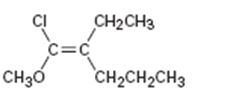
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The two groups attached to the carbons in double bond are to be ranked first. The member that ranks higher can be determined by considering the atomic number of the first atom of the two substituents separately. The atom with highest atomic number gets the higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms away from double bond are considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms. The isomer that has the higher ranked groups on each carbon on the same side of the double bond is said to have Z configuration. If the higher ranked groups are on the opposite sides, the alkene is said to have E configuration.
To assign:
The configuration for the compound given as E or Z.
c)
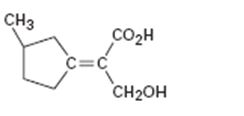
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The two groups attached to the carbons in double bond are to be ranked first. The member that ranks higher can be determined by considering the atomic number of the first atom of the two substituents separately. The atom with highest atomic number gets the higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms away from double bond are considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms. The isomer that has the higher ranked groups on each carbon on the same side of the double bond is said to have Z configuration. If the higher ranked groups are on the opposite sides, the alkene is said to have E configuration.
To assign:
The configuration for the compound given as E or Z.
d)
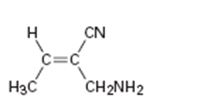
Interpretation:
To assign E or Z configuration for the compound given.
Concept introduction:
The two groups attached to the carbons in double bond are to be ranked first. The member that ranks higher can be determined by considering the atomic number of the first atom of the two substituents separately. The atom with highest atomic number gets the higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms away from double bond are considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms. The isomer that has the higher ranked groups on each carbon on the same side of the double bond is said to have Z configuration. If the higher ranked groups are on the opposite sides, the alkene is said to have E configuration.
To assign:
The configuration for the compound given as E or Z.
Trending nowThis is a popular solution!

Chapter 7 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- H-C- H-C-H H -C-H □B (CH₂)₂C=CH₂ ОС HC-CHCHCH DH,CCH-CHCH, H Q9 Which of the following alkenes exhibits E-Z isomerism? AH,CCH=C(CH₂),arrow_forwardHow to assign E & Z configurations and name tetra-substituted alkenes? And How do you determine tetra-substituted, mono-substituted, di-substituted?arrow_forwardClassify the following alkene as E or Z: CH3 T H3C-CH CI-CH CH3 CH₂Br C-Harrow_forward
- Assign the following alkene molecules as being cis or trans, E or Z isomers.arrow_forwardDraw the structure(s) of all of the alkene isomers, C5H10, that contain a branched chain. Consider E/Z stereochemistry of alkenes.arrow_forwardDraw a structural formula for the product formed upon hydroboration/oxidation of the alkene below. Use wedge and hash bonds ONLY for rings. Do not show stereochemistry in other cases. If the reaction produces a racemic mixture, just draw one stereoisomer.arrow_forward
- 3 In this alkene, highlight the sp²-hybridized carbons in red and the sp³ -hybridized carbons in blue. u Xarrow_forwardQuestion 4arrow_forwardCH3CH2CH2 H CH3CH2CH2 CH3 H2C=CHCH,CH2CH2CH3 CH3 B Which of the alkenes above is the least stable (highest in energy)? Which is the most stable (lowest in energy)?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


