
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 16CI
The compound butyric acid gives rancid butter its characteristics door. (6.7, 7.4,7.5)
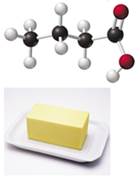
- If black spheres are carbon atoms, white spheres are hydrogen atoms, and red spheres are oxygen atoms, what is the molecular formula of butyric acid?
- What is the molar mass of butyric acid?
- How many grams of butyric acid contain 3.28 × 1023 atoms of oxygen?
- How many grams of carbon are in 5.28 g of butyric acid?
- Butyric acid has a density of 0.959 g/mL at 20?. How many moles of butyric acid are contained in 1.56 mL of butyric acid?
- Identify the bonds C-C, C-H and C-O in a molecule of butyric acid as polar covalent or nonpolar covalent.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Na2SO4 • XH2O
If I have 0.548 g of the hydrate after weighing and 0.272 g of H2O was lost after heating the hydrate, what is the value of x for the hydrate?
A sample of washing soda (a hydrate of sodium carbonate) has the formula Na2CO3.xH2O. When 2.558 –g sample of washing soda is heated at 125 oC, all the water of hydration is lost, leaving 0.948 g of Na2CO3, What is the value of x?
For the following reaction:
5Ca + V2O5 --> 5CaO + 2V
In one process 5 moñ of V2O5 react with 3 mol of acá. Calculate the theoretical yield of V in the unit of mol.
Chapter 8 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 8.1 - Prob. 8.1QAPCh. 8.1 - Prob. 8.2QAPCh. 8.1 - Prob. 8.3QAPCh. 8.1 - Prob. 8.4QAPCh. 8.1 - Prob. 8.5QAPCh. 8.1 - Prob. 8.6QAPCh. 8.1 - Prob. 8.7QAPCh. 8.1 - Prob. 8.8QAPCh. 8.2 - Why do scuba divers need to exhale air when they...Ch. 8.2 - Why does a sealed bag of chips expand when you...
Ch. 8.2 - The air in a cylinder with a piston has a volume...Ch. 8.2 - Prob. 8.12QAPCh. 8.2 - Prob. 8.13QAPCh. 8.2 - Prob. 8.14QAPCh. 8.2 - Prob. 8.15QAPCh. 8.2 - Prob. 8.16QAPCh. 8.2 - Prob. 8.17QAPCh. 8.2 - Prob. 8.18QAPCh. 8.2 - Prob. 8.19QAPCh. 8.2 - Prob. 8.20QAPCh. 8.2 - Prob. 8.21QAPCh. 8.2 - Prob. 8.22QAPCh. 8.3 - Prob. 8.23QAPCh. 8.3 - Prob. 8.24QAPCh. 8.3 - Prob. 8.25QAPCh. 8.3 - Prob. 8.26QAPCh. 8.3 - Prob. 8.27QAPCh. 8.3 - Prob. 8.28QAPCh. 8.4 - Prob. 8.29QAPCh. 8.4 - Prob. 8.30QAPCh. 8.4 - Prob. 8.31QAPCh. 8.4 - Prob. 8.32QAPCh. 8.4 - Prob. 8.33QAPCh. 8.4 - Prob. 8.34QAPCh. 8.5 - Prob. 8.35QAPCh. 8.5 - Prob. 8.36QAPCh. 8.5 - Prob. 8.37QAPCh. 8.5 - Prob. 8.38QAPCh. 8.6 - Prob. 8.39QAPCh. 8.6 - Prob. 8.40QAPCh. 8.6 - Prob. 8.41QAPCh. 8.6 - Prob. 8.42QAPCh. 8.6 - Prob. 8.43QAPCh. 8.6 - Prob. 8.44QAPCh. 8.6 - Prob. 8.45QAPCh. 8.6 - Prob. 8.46QAPCh. 8.7 - Prob. 8.47QAPCh. 8.7 - Prob. 8.48QAPCh. 8.7 - Prob. 8.49QAPCh. 8.7 - Prob. 8.50QAPCh. 8.7 - Prob. 8.51QAPCh. 8.7 - Prob. 8.52QAPCh. 8.7 - Prob. 8.53QAPCh. 8.7 - Prob. 8.54QAPCh. 8.7 - Prob. 8.55QAPCh. 8.7 - Prob. 8.56QAPCh. 8.7 - Prob. 8.57QAPCh. 8.7 - Prob. 8.58QAPCh. 8.7 - Prob. 8.59QAPCh. 8.7 - Prob. 8.60QAPCh. 8.8 - Prob. 8.61QAPCh. 8.8 - Prob. 8.62QAPCh. 8.8 - Prob. 8.63QAPCh. 8.8 - Prob. 8.64QAPCh. 8.8 - Prob. 8.65QAPCh. 8.8 - Prob. 8.66QAPCh. 8.8 - Prob. 8.67QAPCh. 8.8 - Prob. 8.68QAPCh. 8.8 - Prob. 8.69QAPCh. 8.8 - Prob. 8.70QAPCh. 8 - Prob. 8.71UTCCh. 8 - Prob. 8.72UTCCh. 8 - Prob. 8.73UTCCh. 8 - Prob. 8.74UTCCh. 8 - Prob. 8.75UTCCh. 8 - Prob. 8.76UTCCh. 8 - Prob. 8.77UTCCh. 8 - Prob. 8.78UTCCh. 8 - Prob. 8.79AQAPCh. 8 - Prob. 8.80AQAPCh. 8 - Prob. 8.81AQAPCh. 8 - Prob. 8.82AQAPCh. 8 - Prob. 8.83AQAPCh. 8 - Prob. 8.84AQAPCh. 8 - Prob. 8.85AQAPCh. 8 - Prob. 8.86AQAPCh. 8 - Prob. 8.87AQAPCh. 8 - Prob. 8.88AQAPCh. 8 - Prob. 8.89AQAPCh. 8 - Prob. 8.90AQAPCh. 8 - Prob. 8.91AQAPCh. 8 - Prob. 8.92AQAPCh. 8 - Prob. 8.93CQCh. 8 - Prob. 8.94CQCh. 8 - Prob. 8.95CQCh. 8 - Prob. 8.96CQCh. 8 - Prob. 8.97CQCh. 8 - Prob. 8.98CQCh. 8 - Prob. 8.99CQCh. 8 - Prob. 8.100CQCh. 8 - Prob. 8.101CQCh. 8 - Prob. 8.102CQCh. 8 - Prob. 8.103CQCh. 8 - Prob. 8.104CQCh. 8 - Prob. 13CICh. 8 - Prob. 14CICh. 8 - Prob. 15CICh. 8 - The compound butyric acid gives rancid butter its...Ch. 8 - Prob. 17CICh. 8 - Automobile exhaust is a major cause of air...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The active ingredient of Benadryl© Chesty Forte Cough Liquid is Guaiphenesin an expectorant drug used to assist the expectoration ('bringing up') of phlegm from the airways in acute respiratory tract infections. A 200.0 mL bottle of Benadryl© Chesty Forte Cough contains 4.00 g of Guaiphenesin (C10H14O4). If the recommended dose for an adult is 350 mg of Guaiphenesin. How many moles of Guaiphenesin does this equal? (answer to 2 significant figures)arrow_forwarda food contains 131kcal, 24 grams of carbohydrates. What percentage of kcals are from carbohydratesarrow_forwardWhich one of the following statements is false? Group of answer choices -Water is the most important greenhouse gas. -Water plays the greatest role in the thermal regulation of the atmosphere -The amount of energy absorbed when water evaporates is large due to its strong intermolecular forces. -Water molecules absorb energy when water vapor condenses to form clouds.arrow_forward
- Draw a potential energy diagram for a system in which the forward reaction has Eact = +42 kcal/mol and the reverse reaction has Eact = +28 kcal/mol. a. Is the forward process endothermic or exothermic? ["", ""] b. What is the value for ΔH for this reaction? Please insert an image of your workarrow_forwardUSE THE CONDENSED STRUCTURAL FORMULA IN WRITING THE ORGANIC COMPOUND AND GIVE THE NECESSARY COEFFICIENT BESIDE EACH COMPOUND IF NEEDED answer 3 and 4arrow_forwardChoose the most forcing green house gas based on its mass. a) Carbon dioxide b) Methane c) Chloroflurocarbons d) Waterarrow_forward
- a cereal contains 11 grams of sucrose per 60g of cereal. How many grams of cereal must be eated to consume 0.0424 moles of sucrosearrow_forwardA 12 year old male patient has been prescribed Gaviscon suspension 5 mL four times a day. Gaviscon suspension contains 3.1 mmol Na+/5 mL. The recommended daily allowance (RDA) of salt for the patient is 3 g, which is the equivalent to 1.2 g sodium per day. The atomic mass of sodium is 23. What percentage of the patient’s recommended daily salt allowance is contained in his total daily dose of Gaviscon suspension? Answer to the nearest whole number. units - %arrow_forwardIn the 1980s, there was an international agreement to destroy all stockpiles of mustard gas, ClCH2CH2SCH2CH2Cl. When this substance contacts the moisture in eyes, nasal passages, and skin, the −OH groups of water replace the Cl atoms and create high local concentrations of hydrochloric acid, which cause severe blistering and tissue destruction. Write a balanced equation for this reaction and calculate ΔHrxn . Do not include states in your equation.arrow_forward
- 6. A 0.400-g sample of Niobium(IV) oxide is reacted with oxygen gas, yielding 0.4256 g of a new compound, NbxOy. Write a balanced chemical equation for this reaction (include state symbols to indicate whether each compound is solid, gas, liquid, etc.). You may write your coefficients in terms of x and y. What is the molecular formula of the new compound (that is, what are the values of x and y)?arrow_forwarda hydrated CuSO4 salt was heated and thus dehydrated. Determine the % H2O by weight in the hydrated CuSO4 salt. SHOW WORK TO RECEIVE FULL CREDIT mass of empty dish 43.571 gmass of dish + hydrated salt 44.326 gmass of dish + dehydrated salt 44.054 g 4. Using the data found in question number 3, determine the empirical formula for the hydrated CuSO4salt. What is the chemical name of this dehydrated salt? 5. Suppose that after your second heating, you were impatient and did not wait until your porcelain evaporating dish to cool. Instead, you weighed it while it was still hot. How would this specifically affect the calculated percentage of water in the compound?arrow_forwardAcetylene (C2H2) gas is often used in welding torches because of the very high heat produced when it reacts with oxygen (O2) gas, producing carbon dioxide gas and water vapor. Calculate the moles of oxygen needed to produce 1.30 mol of water. Answer with unit symbol and 3 sigarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY