
ORGANIC CHEMISTRY-W/STUD.SOLN.MAN.
10th Edition
ISBN: 9781260001099
Author: Carey
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 8, Problem 65P
On the basis of the mechanism of acid-catalyzed hydration, can you suggest why the reaction
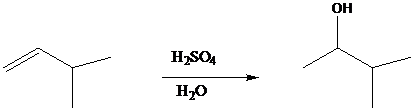
would probably not be a good method for the synthesis of
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Translated version of the problem: Write the obtainment of cinnamic acid (3-phenylpropenoic acid) using benzaldehyde and other necessary reagents, showing the reaction mechanism.
can you solve question 5 ?
Write the obtainment of cinnamic acid (3-phenylpropenoic acid) using benzaldehyde and other necessary reagents, showing the reaction mechanism.
Write a mechanism for the formation of tert-butylbenzene from benzene and tert-butyl alcohol in the presence of phosphoric acid.
Chapter 8 Solutions
ORGANIC CHEMISTRY-W/STUD.SOLN.MAN.
Ch. 8.1 - What three alkenes yield 2-methylbutane on...Ch. 8.2 - Prob. 2PCh. 8.2 - Prob. 3PCh. 8.3 - Prob. 4PCh. 8.4 - Prob. 5PCh. 8.4 - Give a structural formula for the carbocation...Ch. 8.5 - Prob. 7PCh. 8.6 - Instead of the three-step process of Mechanism...Ch. 8.6 - The rates of hydration of the two alkenes shown...Ch. 8.6 - Is the electrophilic addition of hydrogen chloride...
Ch. 8.7 - You can calculate the equilibrium constant for the...Ch. 8.7 - Does the presence or absence of a catalyst such as...Ch. 8.7 - The gas phase reaction of ethanol with hydrogen...Ch. 8.8 - Prob. 14PCh. 8.8 - Hydroborationoxidation of -pinene, like its...Ch. 8.10 - Arrange the compounds 2-methyl-1-butene,...Ch. 8.10 - Give the structure of the product formed when each...Ch. 8.11 - Prob. 18PCh. 8.11 - Prob. 19PCh. 8.12 - Prob. 20PCh. 8.12 - Prob. 21PCh. 8.13 - Prob. 22PCh. 8.14 - Prob. 23PCh. 8.14 - Prob. 24PCh. 8 - How many alkenes yield...Ch. 8 - Prob. 26PCh. 8 - Catalytic hydrogenation of...Ch. 8 - Prob. 28PCh. 8 - Prob. 29PCh. 8 - Prob. 30PCh. 8 - Prob. 31PCh. 8 - A single epoxide was isolated in 7984% yield in...Ch. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - On catalytic hydrogenation over a rhodium...Ch. 8 - Prob. 36PCh. 8 - Prob. 37PCh. 8 - Prob. 38PCh. 8 - Prob. 39PCh. 8 - 1-Butene has a higher heat of hydrogenation than...Ch. 8 - Match the following alkenes with the appropriate...Ch. 8 - The heats of reaction were measured for addition...Ch. 8 - Complete the following table by adding + and -...Ch. 8 - Match the heats of hydrogenation (107 kJ/mol,...Ch. 8 - The iodination of ethylene at 25 C is...Ch. 8 - Specify reagents suitable for converting...Ch. 8 - (a) Which primary alcohol of molecular formula...Ch. 8 - Identify compounds A and B in the retrosynthesis...Ch. 8 - Identify compounds A and B in the retrosynthesis...Ch. 8 - Prob. 50PCh. 8 - On being heated with a solution of sodium ethoxide...Ch. 8 - Compound A (C7H15Br) is not a primary alkyl...Ch. 8 - Prob. 53PCh. 8 - Prob. 54PCh. 8 - A mixture of three alkenes (A, B, and C) was...Ch. 8 - Reaction of 3,3-dimethyl-1-butene with hydrogen...Ch. 8 - Dehydration of 2,2,3,4,4-pentamethyl-3-pentanol...Ch. 8 - Prob. 58PCh. 8 - East Indian sandalwood oil contains a hydrocarbon...Ch. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - On the basis of the mechanism of acid-catalyzed...Ch. 8 - As a method for the preparation of alkenes, a...Ch. 8 - Which of the following is the most reasonable...Ch. 8 - Prob. 68PCh. 8 - Oxymercuration Concerns about mercurys toxicity...Ch. 8 - Prob. 70DSPCh. 8 - Prob. 71DSPCh. 8 - Prob. 72DSPCh. 8 - Prob. 73DSPCh. 8 - Oxymercuration Concerns about mercurys toxicity...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which experimental method would you recommend for the preparation of 1-bromooctane and of t-butyl bromide?arrow_forwardProvide a step by step mechanism for the conversion of cuclohexanol in hot concentrated phosphoric acidarrow_forwardWrite the mechanism of reaction of benzoic acid with thionyl chloridearrow_forward
- Outline a mechanism for the dehydration of 4-methylcyclohexanol catalyzed by phosphoric acid.arrow_forwardWrite the mechanism and show the product of the reaction of 3- pentanone with n-butylamine with the molecule below with a catalytic amount of acid:arrow_forwardWrite the mechanism of the aliphatic nucleophilic substitution reaction between n-butanol and Br- in strongly acidic mediumarrow_forward
- Write a mechanism for the enolization of a ketone. Show how acids and bases can act as catalysists of this reaction.arrow_forwardBased on the mechanism of acid-catalyzed hydration, can you suggest a reason why it is likely that the reaction (shown below) would NOT be a good method for the synthesis of 3-methyl-2-butanol? Use a mechanism for your explanation.arrow_forwardWith the help of a chemical reaction, show the process of complete hydrogenation for 9,12-octadecadienoic acid. What is the side effect and its consequences observed in above processes?arrow_forward
- What would be the product of a reaction involving 2-methyl-2-pentene and HBr both with and without the presence of peroxides?arrow_forwardExplain the purpose of the following steps, write the equation for the rection when applicable. . Using 2,4-dinitrophenylhydrazine and semicarbazide tests to identify aldehydes and ketones.arrow_forwarda) Discuss all the principles that can be used to facilitate acylation reactions. b) Why do aldehydes and ketone undergo addition not substitution reaction? c) What are the disadvantages of carboxylic acid activation using anhydride and also using thionyl chloride (SOCl2) as a reagent?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY