
Concept explainers
Interpretation:
The pH values after the addition of each proportion of the acid to the base is to be determined. Also, the titration curve needs to be drawn.
Concept introduction:
A strong acid or a strong base can completely dissociate into its ions when they are in an aqueous solution. Pyridine is a weak base while hydrochloric acid is a strong acid. When these two chemical species are reacted with each other, they form their ionic forms and an equilibrium state.
Explanation of Solution
Initial pH of the analyte solution; Pyridine is a weak base that forms equilibrium when dissolved in water. The equilibrium is as follows.
The molarity of pyridine is 0.1 M thus, the ICE table can be created as follows:
| Reaction | Pyridine base | Pyridine ion | OH- |
| Initial | 0.1 | 0 | 0 |
| Change | -x | +x | +x |
| Equilibrium | (0.1-x) | x | x |
Here,
Thus,
The pH of the solution will be:
Addition of
Total amount of base to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0004 | -0.0004 | 0.0004 |
| Equilibrium | 0.0021 | 0 | 0.0004 |
In the Henderson-Hasselbalch equation, the pKa is used. Therefore, the pKa for pyridine need to be calculated using its Kb.
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0008 | -0.0008 | 0.0008 |
| Equilibrium | 0.0017 | 0 | 0.0008 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.00125 | -0.00125 | 0.00125 |
| Equilibrium | 0.00125 | 0 | 0.00125 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine base |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.002 | -0.002 | 0.002 |
| Equilibrium | 0.0005 | 0 | 0.002 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0024 | -0.0024 | +0.0024 |
| Equilibrium | 0.0001 | 0 | 0.0024 |
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0.0000 | 0 | 0.0025 |
Concentration of base after addition of base
Concentration of conjugate ion
At this point, there is no excess acid or base. Therefore, the only possible reaction here is the dissociation of the conjugate acid of the pyridine base.
Thereafter, using the Ka value for pyridine, the amount of hydrogen ions in the solution can be determined to get the pH value at this point.
| Reaction | Pyridine base | Pyridine ion | H+ |
| Initial | 0.05 | 0 | 0 |
| Change | X | x | x |
| Equilibrium | (0.05-x) | x | x |
Then the pH can be calculated as follows:
The value of x can be neglected from denominator as the acid dissociation constant has very small value.
Thus,
Addition of 26.0 mL of the acid:
Total amount of pyridine to be neutralized
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0.0026 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0.0000 | 0.0001 | 0.0025 |
The pH of the solution only depends on the concentration of HCl thus,
The pH of the solution will be:
Addition of
Amount of acid added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0.0028 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0 | 0.0003 | 0.0025 |
Here, the pH will only depend on the concentration of hydrogen ion.
Concentration of hydrogen ion
Addition of
Amount of ammonium added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Pyridine base | H+ | Pyridine ion |
| Initial | 0.0025 | 0.0003 | 0 |
| Change | -0.0025 | -0.0025 | +0.0025 |
| Equilibrium | 0 | 0.0005 | 0.0025 |
The pH will only depend on the concentration of hydrogen ion.
Concentration of hydrogen ion
Thus,
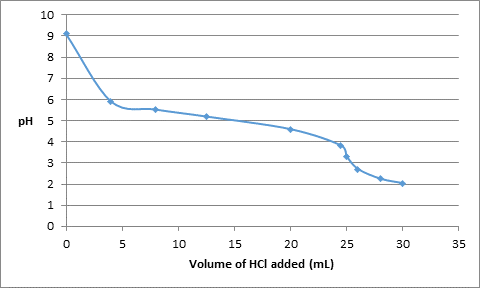
Thus, the data obtained from the calculations is as follows:
| Volume of HCl added (mL) | pH |
| 0 | 9.11 |
| 4.0 | 5.92 |
| 8.0 | 5.53 |
| 12.5 | 5.2 |
| 20.0 | 4.6 |
| 24.5 | 3.82 |
| 25.0 | 3.3 |
| 26.0 | 2.71 |
| 28.0 | 2.25 |
| 30.0 | 2.04 |
The titration curve can be represented as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
Chemical Principles
- Repeat the procedure in Exercise 61, but for the titration of 25.0 mL of 0.100 M pyridine with 0.100 M hydrochloric acid (Kb for pyridine is 1.7 109). Do not calculate the points at 24.9 and 25.1 mL.arrow_forwardThe pH of 0.10 M CH3NH2 (methylamine) is 11.8. When the chloride salt of methylamine, CH3NH3Cl, is added to this solution, does the pH increase or decrease? Explain, using Le Chteliers principle and the common-ion effect.arrow_forwardA solution is made by diluting 25.0 mL of concentrated HCl (37% by weight; density = 1.19 g/mL) to exactly 500 mL. Calculate the pH of the resulting solution.arrow_forward
- Which of the indicators in Fig. 14-8 could be used for the titrations in Exercises 61 and 63?arrow_forwardConsider all acid-base indicators discussed in this chapter. Which of these indicators would be suitable for the titration of each of these? (a) NaOH with HClO4 (b) acetic acid with KOH (c) NH3 solution with HBr (d) KOH with HNO3 Explain your choices.arrow_forwardWhich of the acid-base indicators discussed in this chapter would be suitable for the titration of (a) HNO3 with KOH. (b) KOH with acetic acid. (c) HCl with NH3. (d) KOH with HNO2. Explain your answers.arrow_forward
- 8-93 Do a 1.0 M CH3COOH solution and a 1.0 M HCI solution require the same amount of 1.0 M NaOH to hit a titration end point? Explain.arrow_forwardConsider the titration of 100.0 mL of 0.10 M H3AsO4 by 0.10 M NaOH. What are the major species present at 50.0 mL of NaOH added? How would you calculate the pH at this point? Answer the same questions for 150.0 mL of NaOH added. At what volume of NaOH added does pH = pKa1?arrow_forwardCalculate the pH during the titration of 50.00 mL of 0.100 M Sr(OH)2 with 0.100 M HNO3 after 0, 50.00, 100.00, and 150.00 mL nitric acid have been added. Graph the titration curve and compare with the titration curve obtained in Exercise 16.22.arrow_forward
- A 0.400-M solution of ammonia was titrated with hydrochloric acid to the equivalence point, where the total volume was 1.50 times the original volume. At what pH does the equivalence point occur?arrow_forwardConsider all acid-base indicators discussed in this chapter. Which of these indicators would be suitable for the titration of (a) NaOH with HClO4. (b) acetic acid with KOH. (c) NH3 solution with HBr. (d) KOH with HNO3. Explain your choices.arrow_forwardEstimate the pH that results when the following two solutions are mixed. a) 50 mL of 0.3 M CH3COOH and 50 mL of 0.4 M KOH b) 100 mL of 0.3 M CH3COOH and 50 mL of 0.4 M NaOH c) 150 mL of 0.3 M CH3COOH and 100 mL of 0.3 M Ba(OH)2 d) 200 mL of 0.3 M CH3COOH and 100 mL of 0.3 M Ba(OH)2arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





