
Concept explainers
Interpretation:
The pH values after the addition of each proportion of the base to the acid is to be determined. Also, the titration curve needs to be drawn.
Concept introduction:
Titration curve is drawn to determine the change in pH of an acid or base with respect to the added volume of base or acid to it.
The titration curve can be drawn between a strong/weak acid and strong/weak base. The change in pH shows different patterns for different combinations of acids and bases.
Explanation of Solution
Initial pH of the analyte solution can be calculated as follows:
Lactic acid is a weak acid that forms an equilibrium mixture when dissolved in water. The equilibrium is as follows.
The initial molarity of lactic acid is 0.1 M.
The amount of lactic acid at the beginning can be calculated from. By constructing an ICE table, the concentration of lactate ion in the solution after the acid dissociation can be determined.
| Reaction | Lactic acid | Lactate | H+ |
| Initial | 0.1 | 0 | 0 |
| Change | -x | +x | +x |
| Equilibrium | (0.1-x) | x | x |
The acid dissociation constant can be represented as follows:
Solving this quadratic equation gives the amount of hydrogen ions in the solution.
Thus, the concentration of hydrogen ion is 0.00185 and pH of the solution can be calculated as follows:
Addition of
Total amount of lactic acid to be neutralized can be calculated from its molarity and volume as follows:
Or,
Now, the amount of base added can be calculated as follows:
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.0004 | ||
| Change | -0.0004 | -0.0004 | 0.0004 | 0.0004 |
| Equilibrium | 0.0021 | 0 | 0.0004 | 0.0004 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.0008 | ||
| Change | -0.0008 | -0.0008 | 0.0008 | 0.0008 |
| Equilibrium | 0.0017 | 0 | 0.0008 | 0.0008 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.00125 | ||
| Change | -0.00125 | -0.00125 | 0.00125 | 0.00125 |
| Equilibrium | 0.00125 | 0 | 0.00125 | 0.00125 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.002 | ||
| Change | -0.002 | -0.002 | 0.002 | 0.002 |
| Equilibrium | 0.0005 | 0 | 0.002 | 0.002 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.0024 | ||
| Change | -0.0024 | -0.0024 | 0.0024 | 0.0024 |
| Equilibrium | 0.0001 | 0 | 0.0024 | 0.0024 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.00245 | ||
| Change | -0.00245 | -0.00245 | -0.00245 | -0.00245 |
| Equilibrium | 0.00005 | 0 | -0.00245 | -0.00245 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.00249 | ||
| Change | -0.00249 | -0.00249 | -0.00249 | -0.00249 |
| Equilibrium | 0.00001 | 0 | -0.00249 | -0.00249 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
Applying the Henderson-Hasselbalch equation,
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.0025 | ||
| Change | -0.0025 | -0.0025 | -0.0025 | -0.0025 |
| Equilibrium | 0.0000 | 0 | -0.0025 | -0.0025 |
Concentration of lactic acid after addition of base
Concentration of lactate ion
At this point, there is no excess acid or base. Therefore, the only possible reaction here is the dissociation of the conjugate base of the lactic acid (that is lactate ion).
Thereafter, by obtaining the Kb value for lactate ion, the amount of hydroxide ions in the solution can be determined to get the pH value at this point.
| Reaction | Lactic acid | Lactate | OH- |
| Initial | 0.05 | 0 | 0 |
| Change | -X | x | x |
| Equilibrium | (0.05-x) | x | x |
Then the pH can be calculated as follows:
Thereafter, this quadratic equation can be solved to determine the hydroxide ion concentration, thereby, the pOH and the pH can be determined.
The calculated value of x is concentration of hydroxide ion. The pOH of the solution will be:
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.0028 | ||
| Change | -0.0025 | 0.0025 | 0 | 0 |
| Equilibrium | 0 | 0.0003 | 0 | 0 |
Concentration of hydroxide
Addition of
Total amount of lactic acid to be neutralized
Amount of base added
Then the ICE table after the addition of base is created in order to determine the pH of the solution using Henderson-Hasselbalch equation.
| Reaction | Lactic acid | OH- | Lactate | H+ |
| Initial | 0.0025 | 0 | 0 | 0 |
| Add | 0 | 0.0030 | ||
| Change | -0.0025 | 0.0025 | 0 | 0 |
| Equilibrium | 0 | 0.0005 | 0 | 0 |
Concentration of hydroxide
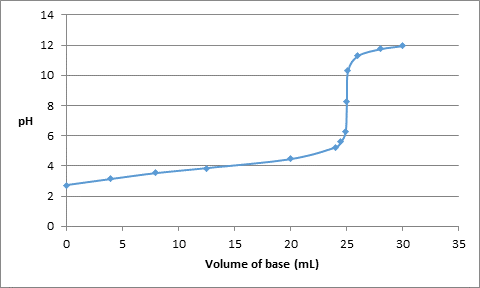
Thus, the value of pH with respect to added volume of base is as follows:
| Volume (in mL) | pH |
| 0 | 2.73 |
| 4 | 3.14 |
| 8 | 3.53 |
| 12.5 | 3.86 |
| 20 | 4.46 |
| 24 | 5.24 |
| 24.5 | 5.6 |
| 24.9 | 6.3 |
| 25.0 | 8.28 |
| 25.1 | 10.3 |
| 26.0 | 11.30 |
| 28.0 | 11.75 |
| 30.0 | 11.96 |
The titration curve can be drawn as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
Chemical Principles
- Consider the titration of 100.0 mL of 0.10 M H3AsO4 by 0.10 M NaOH. What are the major species present at 50.0 mL of NaOH added? How would you calculate the pH at this point? Answer the same questions for 150.0 mL of NaOH added. At what volume of NaOH added does pH = pKa1?arrow_forwardA buffer is prepared by mixing 525 mL of 0.50 M formic acid, HCHO2, and 475 mL of 0.50 M sodium formate, NaCHO2. Calculate the pH. What would be the pH of 85 mL of the buffer to which 8.6 mL of 0.15 M hydrochloric acid had been added?arrow_forwardFor the titration of 50.0 mL of 0.150 M ethylamine. C2H5NH2, with 0.100 M HCl, find the pH at each of the following points, and then use that information to sketch the titration curve and decide on an appropriate indicator. (a) At the beginning, before HCl is added (b) At the halfway point in the titration (c) When 75% of the required acid has been added (d) At the equivalence point (e) When 10.0 mL more HCl has been added than is required (f) Sketch the titration curve. (g) Suggest an appropriate indicator for this titration.arrow_forward
- Ka for formic acid is 1.7 104 at 25C. A buffer is made by mixing 529 mL of 0.465 M formic acid, HCHO2, and 494 mL of 0.524 M sodium formate, NaCHO2. Calculate the pH of this solution at 25C after 110 mL of 0.152 M HCl has been added to this buffer.arrow_forwardLactic acid is a common by-product of cellular respiration and is often said to cause the burn associated with strenuous activity. A 25.0-mL sample of 0.100 M lactic acid (HC3H5O3, pKa = 3.86) is titrated with 0.100 M NaOH solution. Calculate the pH after the addition of 0.0 mL, 4.0 mL, 8.0 mL, 12.5 mL, 20.0 mL, 24.0 mL, 24.5 mL, 24.9 mL, 25.0 mL, 25.1 mL, 26.0 mL, 28.0 mL, and 30.0 mL of the NaOH. Plot the results of your calculations as pH versus milliliters of NaOH added.arrow_forwardAniline hydrochloride, (C6H5NH3)Cl, is a weak acid. (Its conjugate base is the weak base aniline, C6H5NH2.) The acid can be titrated with a strong base such as NaOH. C6H5NH3+(aq)+OH(aq)C6H5NH2(aq)+H2O(l) Assume 50.0 mL of 0.100 M aniline hydrochloride is titrated with 0.185 M NaOH. (Ka for aniline hydrochloride is 2.4 105.) (a) What is the pH of the (C6H5NH3) solution before the titration begins? (b) What is the pH at the equivalence point? (c) What is the pH at the halfway point of the titration? (d) Which indicator in Figure 17.11 could be used to detect the equivalence point? (e) Calculate the pH of the solution after adding 10.0, 20.0, and 30.0 mL of base. (f) Combine the information in parts (a), (b), (c), and (e), and plot an approximate titration curve.arrow_forward
- Find the pH of the solution obtained when 25 mL of 0.065 M benzylamine, C7H7NH2, is titrated to the equivalence point with 0.050 M hydrochloric acid. Kb for benzylamine is 4.7 1010.arrow_forwardWhen 40.00 mL of a weak monoprotic acid solution is titrated with 0.100-M NaOH, the equivalence point is reached when 35.00 mL base has been added. After 20.00 mL NaOH solution has been added, the titration mixture has a pH of 5.75. Calculate the ionization constant of the acid.arrow_forward2. If an acetic acid/sodium acetate buffer solution is prepared from 100. mL of 0.10 M acetic acid what volume of 0.10 M sodium acetate must be added to have a pH of 4.00? 100. mL 50. mL 36 mL 18 mLarrow_forward
- A 0.288-g sample of an unknown monoprotic organic acid is dissolved in water and titrated with a 0.115 M sodium hydroxide solution. After the addition of 17.54 mL of base a pH of 4.92 is recorded. The equivalence point is reached when a total of 33.83 mL of NaOH is added. a What is the molar mass of the organic acid? b What is the Ka value for the acid? The Ka value could have been determined very easily if a pH measurement had been made after the addition of 16.92 mL of NaOH. Why?arrow_forwardWhen a diprotic acid, H2A, is titrated with NaOH, the protons on the diprotic acid are generally removed one at a time, resulting in a pH curve that has the following generic shape: a. Notice that the plot has essentially two titration curves. If the first equivalence point occurs at 100.0 mL NaOH added, what volume of NaOH added corresponds to the second equivalence point? b. For the following volumes of NaOH added, list the major species present after the OH reacts completely. i. 0 mL NaOH added ii. between 0 and 100.0 mL NaOH added iii. 100.0 mL NaOH added iv. between 100.0 and 200.0 mL NaOH added v. 200.0 mL NaOH added vi. after 200.0 mL NaOH added c. If the pH at 50.0 mL NaOH added is 4.0, and the pH at 150.0 mL NaOH added is 8.0, determine the values Ka1, and Ka2 for the diprotic acid.arrow_forwardCalculate the pH change when 10.0 mL of 0.100-M NaOH is added to 90.0 mL pure water, and compare the pH change with that when the same amount of NaOH solution is added to 90.0 mL of a buffer consisting of 1.00-M NH3 and 1.00-M NH4Cl. Assume that the volumes are additive. Kb of NH3 = 1.8 × 10-5.arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





