
Package: Organic Chemistry With Connect 2-semester Access Card
4th Edition
ISBN: 9781259671838
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 8.12P
What
a.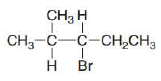 b.
b.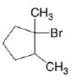 c.
c.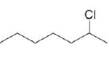 d.
d.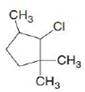
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2
Devise a synthesis of each alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents.
Draw all possible constitutional isomers formed by dehydrohalogenation of each alkyl halide.
Chapter 8 Solutions
Package: Organic Chemistry With Connect 2-semester Access Card
Ch. 8 - Label the and carbons in each alkyl halide. Draw...Ch. 8 - Problem 8.2 Classify each alkene in the following...Ch. 8 - Prob. 8.3PCh. 8 - Prob. 8.4PCh. 8 - Problem 8.5 Label each pair of alkenes as...Ch. 8 - Prob. 8.6PCh. 8 - Problem 8.7 Several factors can affect alkene...Ch. 8 - Prob. 8.8PCh. 8 - Prob. 8.9PCh. 8 - Prob. 8.10P
Ch. 8 - Prob. 8.11PCh. 8 - What alkenes are formed from each alkyl halide by...Ch. 8 - Prob. 8.13PCh. 8 - Prob. 8.14PCh. 8 - Problem 8.15 How does each of the following...Ch. 8 - Prob. 8.16PCh. 8 - Prob. 8.17PCh. 8 - Prob. 8.18PCh. 8 - Prob. 8.19PCh. 8 - Problem 8.19 Explain why...Ch. 8 - Prob. 8.21PCh. 8 - Draw the alkynes formed when each dihalide is...Ch. 8 - Prob. 8.23PCh. 8 - Draw a stepwise mechanism for the following...Ch. 8 - 8.24 Rank the alkenes shown in the ball-and-stick...Ch. 8 - Prob. 8.26PCh. 8 - 8.26 What is the major E2 elimination product...Ch. 8 - Prob. 8.28PCh. 8 - Prob. 8.29PCh. 8 - Prob. 8.30PCh. 8 - Prob. 8.31PCh. 8 - PGF2 is a prostaglandin, a group of compounds that...Ch. 8 - Prob. 8.33PCh. 8 - Prob. 8.34PCh. 8 - Prob. 8.35PCh. 8 - Prob. 8.36PCh. 8 - Prob. 8.37PCh. 8 - Prob. 8.38PCh. 8 - Prob. 8.39PCh. 8 - Prob. 8.40PCh. 8 - What alkene is the major product formed from each...Ch. 8 - Prob. 8.42PCh. 8 - Prob. 8.43PCh. 8 - Prob. 8.44PCh. 8 - 8.42 In the dehydrohalogenation of...Ch. 8 - Prob. 8.46PCh. 8 - Prob. 8.47PCh. 8 - Prob. 8.48PCh. 8 - Prob. 8.49PCh. 8 - Prob. 8.50PCh. 8 - Draw the products of each reaction. a.c. b.d.Ch. 8 - Draw the structure of a dihalide that could be...Ch. 8 - Under certain reaction conditions, 2,...Ch. 8 - For which reaction mechanisms, SN1, SN2, E1 or...Ch. 8 - Draw the organic products formed in each reaction....Ch. 8 - Prob. 8.56PCh. 8 - Prob. 8.57PCh. 8 - Draw all products, including stereoisomers, in...Ch. 8 - Draw all of the substitution and elimination...Ch. 8 - Prob. 8.60PCh. 8 - 8.59 Draw a stepwise, detailed mechanism for each...Ch. 8 - Draw the major product formed when...Ch. 8 - Draw a stepwise, detailed mechanism for the...Ch. 8 - Explain why the reaction of with gives ...Ch. 8 - Draw a stepwise detailed mechanism that...Ch. 8 - Prob. 8.66PCh. 8 - 8.65 Explain the selectivity observed in the...Ch. 8 - Prob. 8.68PCh. 8 - Although dehydrohalogenation occurs with anti...Ch. 8 - 8.68 (a) Draw all products formed by treatment of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (a) What is the major alkene formed when A is dehydrated with H2SO4? (b) What is the major alkene formed when A is treated with POCl3 and pyridine? Explain why the major product is different in these reactions.arrow_forwardFill in the missing reagents a-h in the following scheme:arrow_forwardWhat reagents would you use to prepare each of the following from 3-hexene? a. b.CH3CH2CH2CH2CH2CH3 c. d.arrow_forward
- What Wittig reagent and carbonyl compound are needed to prepare each alkene? When two routes are possible, indicate which route, if any, is preferred.arrow_forwardWhat alkenes are formed from each alkyl halide by an E2 reaction? Use the Zaitsev rule to predict the major product.arrow_forwardDraw a stepwise mechanism for the following substitution. Explain why 2-chloropyridine reacts faster than chlorobenzene in this type of reaction.arrow_forward
- What starting materials are needed to prepare each alkene by a Wittig reaction? When there are two possible routes, indicate which route, if any, is preferred.arrow_forwardβ-D-Glucose, a hemiacetal, can be converted to a mixture of acetals on treatment with CH3OH in the presence of acid. Draw a stepwise mechanism for this reaction. Explain why two acetals are formed from a single starting material.arrow_forwardLabel the α and β carbons in attached alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+−OC(CH3)3.arrow_forward
- For alkenes A, B, C, and D: (a) Rank A—D in order of increasing heat of hydrogenation; (b) rank A—D in order of increasing rate of reaction with H2, Pd-C; (c) draw the products formed when each alkene is treated with ozone, followed by Zn, H2O.arrow_forwardDimethyl cyclopropanes can be prepared by the reaction of an α,β-unsaturated carbonyl compound X with two equivalents of a Wittig reagent Y. Draw a stepwise mechanism for this reaction.arrow_forwardDimethyl cyclopropanes can be prepared by the reaction of an α, β- unsaturated carbonyl compound X with two equivalents of a Wittig reagent Y. Draw a stepwise mechanism for this reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY