
Concept explainers
Draw the structure of the conjugate base of each acid.
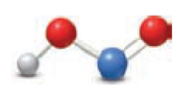
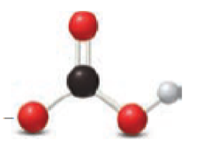
(a)
Interpretation:
The conjugate base of the acid
Concept Introduction:
Conjugate base: A conjugate base is the product formed by a loss of proton from an acid. The conjugate base of the acid A will be
Explanation of Solution
A proton is removed from the acid to form the conjugate base.
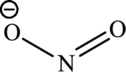
Nitrous acid loses a proton to form nitrite ion
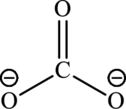
(b)
Interpretation:
The conjugate base of carbonic acid has to be drawn.
Concept Introduction:
Refer to part (a).
Explanation of Solution
The conjugate base is formed by removing a proton from the acid.
Carbonic acid
Want to see more full solutions like this?
Chapter 8 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Several acids and their respective equilibrium constants are: Which is the strongest acid? Which is the weakest acid? Which acid has the weakest conjugate base? Which acid has the strongest conjugate base?arrow_forwardMark each of the following statements True or False: a. The conjugate base of a strong acid is always a weak base. b. The conjugate acid of a strong base is always a weak acid. c. The stronger the acid, the weaker its conjugate base, and vice versa.arrow_forwardExplain the difference between a strong acid and a weak acid.arrow_forward
- Which of the following compounds or ions has the weakest conjugate base? Briefly explain your choice. a) HCN b) HClO c) NH4+arrow_forwardClassify each compound as an Arrhenius acid or an Arrhenius base H2S RbOH Mg(OH)2 H3PO4arrow_forwardSolution A has a pH of 2.0. Solution B has a pH of 5.0 Which solution is more acidic? Based on the H+ ion concentrations in the two solutions, how many times more acidic?arrow_forward
- Consider the following four solutions: (1) apple juice, pH 3.8, (2) pickle juice, pH 3.5, (3) carbonated beverage, pH 3.0, and (4) drinking water, pH 7.2. a. Which solution has the highest [H3O+]? b. Which solution has the highest [OH]? c. List the solutions in order of increasing acidity. d. List the solutions in order of decreasing basicity.arrow_forwardHow do the components of a conjugate acid—base pair differ from one another4? Give an example of a conjugate acid—base pair to illustrate your answer.arrow_forwardList the following bases in order of their decreasing strength strongest base first: CN,H2O,HSO3,ClO,Cl.arrow_forward
- Answer true or false to the following statements about the mechanism of acid-base reactions. (a) The acid and base must encounter each other by a collision in order for the proton to transfer. (b) All collisions between acids and bases result in proton transfer. (c) During an acid-base reaction the lone pair on the base fills the A-H antibonding sigma orbital.arrow_forwardConsider the following four biological solutions: (1) bile, pH 8.0, (2) blood, pH 7.4, (3) urine, pH 6.0, and (4) gastric juice, pH 1.6. a. Which solution has the lowest [H3O+]? b. Which solution has the lowest [OH]? c. List the solutions in order of decreasing acidity. d. List the solutions in order of increasing basicity.arrow_forwardPure liquid ammonia ionizes in a manner similar to that of water. (a) Write the equilibrium for the autoionization of liquid ammonia. (b) Identify the conjugate acid form and the base form of the solvent. (c) Is NaNH2 an acid or a base in this solvent? (d) Is ammonium bromide an acid or a base in this solvent?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





