
Concept explainers
For which reaction mechanisms,
a. The mechanism involves carbocation intermediates.
b. The mechanism has two steps.
c. The reaction rate increases with better leaving groups.
d. The reaction rate increases when the solvent is changed from
e. The reaction rate depends on the concentration of the alkyl halide only.
f. The mechanism is concerted.
g. The reaction of
h. Racemization of a stereogenic center occurs.
i. Tertiary
j. The reaction follows a second-order rate equation.
(a)
Interpretation: The validation of the given statement is to be stated according to the mechanism involved in the reaction.
Concept introduction: The two-step unimolecular elimination reaction that favors the removal of a HX substituent and the formation of a carbocation intermediate takes place in its first step. In the second step of the reaction, the carbocation forms a double bond. This type of reaction is termed E1 elimination reaction.
The two-step unimolecular reaction which favors the removal of a HX substituent and the formation of a carbocation intermediate takes place in its first step. Then, in the second step, the carbocation undergoes substitution. This type of reaction is termed as
Answer to Problem 8.53P
The given statement is true for
Explanation of Solution
The two-steps reactions are
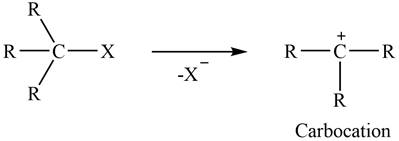
Figure 1
(b)
Interpretation: The reaction in which the mechanism has two steps is to be stated.
Concept introduction: The two-step unimolecular elimination reaction that favors the removal of a HX substituent and the formation of a carbocation intermediate takes place in its first step. In the second step of the reaction, the carbocation forms a double bond. This type of reaction is termed
The two-step unimolecular reaction which favors the removal of a HX substituent and the formation of a carbocation intermediate takes place in its first step. Then, in the second step, the carbocation undergoes substitution. This type of reaction is termed as
Answer to Problem 8.53P
Explanation of Solution
The two-steps reactions are
(c)
Interpretation: The reactions for which the reaction rate increases with better leaving groups is to be stated.
Concept introduction: A good leaving groups is preferred by all substitution and elimination reactions as they increase the rate of the reaction by lowering the transition state energy.
Answer to Problem 8.53P
The rate of
Explanation of Solution
The more the bigger the atom the better it will be as a leaving group and more efficiently it will leave the group to which they are attached and hence increasing the reaction rate. They do so by lowering the transition state energy.
The rate of
(d)
Interpretation: The reactions for which the reaction rate increases when the solvent is changed from
Concept introduction: The choice of the solvent being used determines the mechanism being followed.
Answer to Problem 8.53P
The rate of
Explanation of Solution
The rate of
(e)
Interpretation: The reactions for which the reaction rate depends on the concentration of the alkyl halide only is to be stated.
Concept introduction: The rate law for
The rate law depends only on the concentration of alkyl halide.
Answer to Problem 8.53P
For
Explanation of Solution
The two step reactions are
The rate of both the reactions depends only on the concentration of alkyl halide and is independent of the base concentration.
For
(f)
Interpretation: The reaction for which the mechanism is concerted is to be stated.
Concept introduction: The one-step bimolecular elimination reaction that favors the removal of a proton from carbon adjacent to the leaving group by a base that results in the formation of a carbocation. Then, the formation of a double bond takes place simultaneously. This type of reaction is termed
The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Answer to Problem 8.53P
Explanation of Solution
In a concerted mechanism, all the bond breaking and bond making takes place simultaneously in a single step. In
(g)
Interpretation: The mechanism followed by the reaction of
Concept introduction: Substitution and elimination depends on the base being employed.
Answer to Problem 8.53P
Explanation of Solution
In the given question the alkyl halide is primary and the base is a strong base as well as a strong nucleophile. So, substitution as well as elimination is possible. Since,
(h)
Interpretation: The reaction for which racemization of a stereogenic center occurs is to be stated.
Concept introduction: Racemization is possible where a planar carbocation is the intermediate.
Answer to Problem 8.53P
For SN1 reaction, racemization of a stereogenic center occurs.
Explanation of Solution
Racemization can take place where a planar carbocation intermediate is formed and the incoming nucleophile approaches it from both the sides. In a SN1 reaction, a planar carbocation is formed as the intermediate and attack of the nucleophile is possible from both the sides leading to a mixture of enantiomers being formed.
For
(i)
Interpretation: The reactions for which tertiary
Concept introduction: A tertiary carbocation is more stable then secondary followed by a primary carbocation.
Answer to Problem 8.53P
For
Explanation of Solution
In SN1 and E1 reaction, the first step is the formation of carbocation and follows the order
For
(j)
Interpretation: The reaction which follows a second-order rate equation is to be stated.
Concept introduction: Second order reaction is one in which the reaction rate depends on the concentration of both the reacting species.
Answer to Problem 8.53P
Explanation of Solution
In
Want to see more full solutions like this?
Chapter 8 Solutions
ORG.CHEMISTRY CONNECT ACCESS>CUSTOM<
Additional Science Textbook Solutions
Organic Chemistry
Chemistry by OpenStax (2015-05-04)
EBK INTRODUCTION TO CHEMISTRY
Fundamentals of Heat and Mass Transfer
Introduction to Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
- C3H6 + H2O + H2SO4 --> C3H8O + H2SO4 what is the catalyst in the reaction above?arrow_forwardAn SN1 reaction is shown in the box; the reaction energy profile diagram for this reaction is shown below. Identify the location for Compound I on the reaction profile. A) 1 B) 2 C) 3 D) 4 E) 5arrow_forwardFor which reaction mechanisms—SN1, SN2, E1, or E2—are each of the following statements true? A statement may be true for one or more mechanisms.a. The mechanism involves carbocation intermediates.b. The mechanism has two steps.c. The reaction rate increases with better leaving groups.d. The reaction rate increases when the solvent is changed from CH3OH to (CH3)2SO.e. The reaction rate depends on the concentration of the alkyl halide only.f. The mechanism is concerted.g. The reaction of CH3CH2Br with NaOH occurs by this mechanism.h. Racemization at a stereogenic center occurs.i. Tertiary (3°) alkyl halides react faster than 2° or 1° alkyl halides.j. The reaction follows a second-order rate equation.arrow_forward
- In the reaction observed in part III, where is the slow step? do you expect the reaction in part III to be faster for tert-butyl chloride or 2-chloropropane and why? Part III Silver Nitrate Test for Tertiary Alkyl Halides Set up the reaction. Obtain three 13 100 test tubes and label 13. Pipet 10 drops of distilled water into Test Tube 1. Pipet 10 drops of tert-butyl chloride into Test Tube 2 Pipet 10 drops of your product into Test Tube 3. Add approximately 1 mL of the 1% ethanolic silver nitrate solution to each test tube. Swirl each test tube to mix the contents. The appearance of a white precipitate indicates the presence of a 3° halide. Record your observations in the data table. Note: Avoid getting the AgNO3 solution on your skin. At the end of the experiment, discard the solutions as directed by your instructor. IMAGES PROVIDEDarrow_forwardTrue or false 1.an intermediate is produced in the SN2 reactiom is a carboncation. 2.In SN1 reactions nucleophile HI will react faster than HCL 3.Halogenated waste can be disposed off in a general waste solvent.arrow_forward10 What is the rate of reaction when the transition state free energy is 22.84 kJ/mol? What is the rate of reaction when the transition state free energy is 142 kJ/mol? SHOW YOUR WORK.arrow_forward
- Please explain why reagent 1 cannot be (Br2, FeBr3) 1. Identify reagent 1, and draw the mechanism & product in this step. 2. Identify reagnet 2 (answer given in the picture), and draw the mechanism & product in this step. 3. Explain why Reagent 3 is Zn(Hg), HCl ? draw the mechanism of what happen to the compound when Reagent 3 is Zn(Hg), HCl is added.arrow_forwardWhat is the rate-determining step for an E1 reaction mechanism?arrow_forward8.29 Give the mechanistic symbols (Sn1, Sn2, E1, E2) that are most consistent with each of the following statements: (g) Reactions proceeding by these mechanisms are stereospecific (h) These reaction mechanisms involve carbocation intermediates (i) These reaction mechanisms are the ones most likely to have been involved when the products are found to have a different carbon skeleton from the substratearrow_forward
- Assignment 4 - Energy Diagram Illustrating Your Experience of Snowmaggedon The instructions are simple, draw an energy diagram (or reaction coordinate diagram) that illustrates your experience during our snowmaggedon (think Sn1/Sn2/E1/E2 reaction coordinate diagram). The diagram is to illustrate your "reaction" progress of your experience as you navigated this week. Add as many transition states and intermediates that you see fit (be sure to give detail of each hill and valley). For example: What was the highest, lowest, moderate transition state during your reaction and why? Was your experience exothermic or endothermic? (draw accordingly (-))arrow_forwardA carbocation intermediate is central to which of these reaction types? E1, E2, SN1, SN2 ? Choose 1arrow_forwardWhat are all three products that can result from this E1 mechanism? (1 major, 2 minor)?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


