
Interpretation: The electron dot structure for the methane molecule needs to be drawn. The two molecular orbital pictures for methane molecules need to be drawn as per the given information in the question. The geometry of the methane molecule for each sketch needs to be determined using a simple overlap method. The preferable picture needs to be identified based on the fact that H-C-H bond angles in methane are
Concept Introduction: An electron dot structure represents the arrangement of total valence electrons in a molecule. Here, electrons are represented as dots in pairs around the symbol of atoms of the molecule.
VSEPR theory is used to predict the molecular shape with the help of electron pairs around the central atoms of the molecule. It is based on the assumption that the shape of the molecule is such that there is minimum electronic repulsion between the valence shell and the atom.
Explanation of Solution
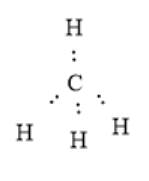
In a methane molecule, one carbon atom is bonded with 4 hydrogen atoms. Here, all the C-H bonds are single. The number of valence electrons on the C atom is 4; thus, central atom C does not have any lone pair of electrons. The electron dot structure can be represented as follows:
According to the question, in the first molecular orbital picture of methane, one electron from
orbital. Here, each half-filled atomic orbital of the carbon atom is overlapped with the half-filled orbital of hydrogen. The molecular orbital diagram can be represented as follows:
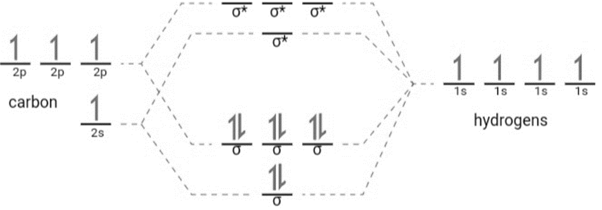
By simple overlap method, the geometry of the methane molecule can be determined as follows:
The number of bonding and non-bonding pair of electrons is 4. Thus, the central atom C is
As per the second sketch, hybridization of 2s and 2p orbitals of carbon is assumed. In this case, the geometry of the molecule will not be tetrahedral as the carbon atom will not be
Since all the C-H bond distances are identical and the bond angle of the H-C-H bond is 109.5 degrees, the preferred sketch will be the first one where it has tetrahedral geometry.
Chapter 8 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





