
(a)
Interpretation: The major product should be determined when the following reactant (alkene) treated with
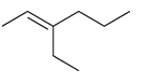
Concept introduction:
(b)
Interpretation: The major product should be determined when the following reactant (alkene) treated with
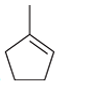
Concept introduction:
Alkenes are unsaturated hydrocarbons. Due to the presence of a double bond, alkenes undergo addition reactions. Some examples of addition reactions of alkenes are hydrohalogenation, halogenation, catalytic hydrogenation, acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc.
(c)
Interpretation: The major product should be determined when the following reactant (alkene) treated with
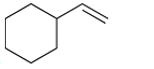
Concept introduction:
Alkenes are unsaturated hydrocarbons. Due to the presence of a double bond, alkenes undergo addition reactions. Some examples of addition reactions of alkenes are hydrohalogenation, halogenation, catalytic hydrogenation, acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc.
(d)
Interpretation: The major product should be determined when the following reactant (alkene) treated with
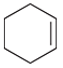
Concept introduction:
Alkenes are unsaturated hydrocarbons. Due to the presence of a double bond, alkenes undergo addition reactions. Some examples of addition reactions of alkenes are hydrohalogenation, halogenation, catalytic hydrogenation, acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY,SOLNS...-ETEXT+BOX
- When pent-1-yne is reacted with BH3 and then with H₂O2, NaOH, which of the following is the major product? H OH en HOarrow_forwardThe following product can be synthesized from the reaction of an alkene with Hg(OAc)2 followed by reduction with NaBH4. Draw all possible alkenes that will give the product shownarrow_forwardDraw the starting structure that would lead to the major product shown under the provided conditions. Drawing H3O+ HOarrow_forward
- s the major organic product obtained from the following reaction? 3 4 2 ○ 1 H₂NNH₂ NH 1 NNH2 3 NH2 2 NHNH2arrow_forwardWhat is the expected major product of reacting cyclohexane carbaldehyde with: 1) CH3MgBr 2) H3O+arrow_forwardIdentify two pairs of an alkyl bromide and an alkene that could be used in a Heck reaction to prepare the following compound:arrow_forward
- Give the major product(s) for each step of the following sequence of reactions:arrow_forwardDetermine a stepwise mechanism for the following reaction that illustrates why two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under Syl reaction conditions, even though it is a 1° alkyl halide. 1-bromohex-2-ene Part 1: Br CH₂OH The first step in the reaction proceeds according to which mechanism? CH₂CH₂CH₂CH=CHCH₂ + CH₂OH CH₂CH₂CH₂CH: Part 2: Br CHCH₂ Draw the missing resonance contributor. OCH3 + CH3CH₂CH₂CH=CHCH₂ CH3CH₂CH₂CH=CH-CH₂ + Br Br OCH 3 H₂CH₂CH₂CH=CH₂ view structure + + Br HBr Xarrow_forwardWhat is the major organic product obtained from the following reaction? CH3 HNO3 H₂SO4 CH3 CH₂NH2 NO₂ CH3 NO2 6866 1 2 3 3 B2 1 4 NH2arrow_forward
- Consider the following carbocation: Which of the following carbocations would result from its rearrangement? le lo A ΟΑ OB Save for Later > m B C Darrow_forwardWhich of the following reagents will react with 2,6-dimethyl-3-octyne to give (E)-2,6-dimethyl-oct-3-ene? H2 / Pd Na / NH3 Lindlar's catalyst NaHarrow_forwardOH oth -H H H3C- NX XT OAc ACO -OAc & O Dess-Martin periodinane provides a means to oxidize a primary alcohol to an aldehyde, while avoiding the use of toxic Cr(VI) compounds. The mechanism involves a reaction similar to the E2 elimination, whereby a C=O double bond is formed with a reduced iodine compound as the leaving group. The reaction occurs in a nonaqueous solvent, CH₂Cl₂, so that the aldehyde product can be recovered. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions H3C CH3 H ACO $$ H H3C H3C H :0: H H3C OH OAC чьarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





