
Concept explainers
(a)
Interpretation: The reaction of an alkene with dilute sulphuric acid in the presence of methanol as solvent occurs quite similar to acid-catalyzed hydration but in the second step alcohol acts as a nucleophile. The mechanism for the given chemical transformation is to be interpreted.
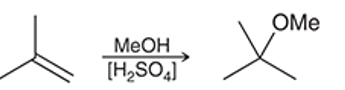
Concept introduction:
(b)
Interpretation: The mechanism for the given chemical transformation is to be interpreted.
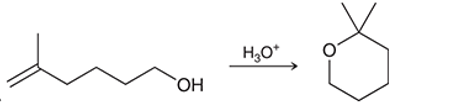
Concept introduction:
Alkenes are unsaturated hydrocarbons with at least one double bond between the carbon atoms. The acid-catalyzed hydration reactions of alkene involve the reaction of an alkene with
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY,SOLNS...-ETEXT+BOX
- The following molecule is an intermediate in the sythesis of strychnine, a poison commonly used as a rodenticide. The intermediate has a free alkoxide group, which serves as a nucleophile. Which site is electrophilic and in close enough proximity to be attacked by this nucleophile?arrow_forwardDraw the reaction mechanism for the reaction between an aldehyde and water under acidic conditions. (H') H,0arrow_forwardDraw a stepwise mechanism for the following reaction sequence.arrow_forward
- Hello, I do not understand these questions and I am stuck. May I get help please?? Question: Draw the curved arrow mechanism for the following reactionarrow_forwardOn a scrap piece of paper, draw a curved arrow mechanism for the following reaction. Once you have determined the major product, draw it in the space provided below. NH₂ + CH₂O, HCl (cat.)arrow_forward1) Please draw the mechanism for the following transformation. OH NABH, ELOHarrow_forward
- Provide a mechanism for the following reaction and draw the product.arrow_forwardIn a Wittig reaction, a ketone or aldehyde reacts as an electrophile with a nucleophile called a Wittig reagent (or phosphonium ylide) to produce an alkene. The Wittig reagent is commonly synthesized first in a two-step process beginning with an alkyl halide, then reacted with the carbonyl compound. In this problem, you'll explore the mechanism of a multi-step synthesis to make an alkene using the Wittig approach.arrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forward
- Draw a curved arrow mechanism that explains the formation of the given organic product in the following reaction. You may need to re-draw structures to show bonds or lone pairs. : ÖH H₂SO4 to 0 :0 2 + 10 And/Remove step Xarrow_forwardDraw the mechanism for this reaction.arrow_forwardPlease explain in detail idk how to go about this problem.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

