
ORGANIC CHEMISTRY,SOLNS...-ETEXT+BOX
4th Edition
ISBN: 9781119760702
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 8.8, Problem 21ATS
Interpretation Introduction
Interpretation: The structure of both the product and major diastereomer should be determined based on the below reaction.
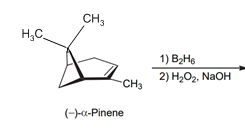
Concept introduction:
The acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc. are some examples of addition reactions of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Sharpless epoxidation can be used to stereoselectively convert an allylic alcohol to an
epoxy alcohol using Ti(O/Pr)4, t-butyl hydroperoxide (TBHP) and a chiral diethyl tartrate
(DET), as exemplified in the formation of compound E below.
HO
OBn
Ti(O/Pr)4
TBHP
L-(+)-DET
CH₂Cl₂
HO
E
OBn
(i) Assign the configuration of the two chiral centres in compound E using CIP rules.
(ii)
E is produced in 94% ee under the conditions. What is the percentage of each
enantiomer?
(iii)
E can be readily debenzylated to give a diol structure, is the diol chiral? Explain.
G.311.
Dehydrohalogenation of vinyl halides is essentially an E2 process. A stercochemical study tevealed that (Z)-2-chloro-2-butendioic acid reacted 50 times faster than its E
stereoisomer.
1. Na+ NH2
HO,C-C=c-co;H
2. H,0
COH
For the reaction below
CH,CH,
1. Na NH2
2. H,0
Draw the structure of the major organic product.
Chapter 8 Solutions
ORGANIC CHEMISTRY,SOLNS...-ETEXT+BOX
Ch. 8.3 - Provide a systematic name for each of the...Ch. 8.3 - Prob. 2CCCh. 8.3 - Prob. 3CCCh. 8.3 - Prob. 4CCCh. 8.5 - Prob. 5CCCh. 8.5 - Prob. 6CCCh. 8.5 - Prob. 1LTSCh. 8.5 - Prob. 7PTSCh. 8.5 - Prob. 8ATSCh. 8.5 - Prob. 9CC
Ch. 8.5 - Prob. 2LTSCh. 8.5 - Prob. 10PTSCh. 8.5 - Prob. 11ATSCh. 8.6 - Prob. 12CCCh. 8.6 - Prob. 13CCCh. 8.6 - Prob. 3LTSCh. 8.6 - Prob. 14PTSCh. 8.6 - Prob. 15ATSCh. 8.7 - Predict the product for each reaction, and predict...Ch. 8.7 - Prob. 17CCCh. 8.8 - Prob. 18CCCh. 8.8 - Prob. 19CCCh. 8.8 - Prob. 4LTSCh. 8.8 - Prob. 20PTSCh. 8.8 - Prob. 21ATSCh. 8.9 - Prob. 5LTSCh. 8.9 - Prob. 22PTSCh. 8.9 - Prob. 23ATSCh. 8.10 - Prob. 24CCCh. 8.10 - Prob. 6LTSCh. 8.10 - Prob. 25PTSCh. 8.10 - Prob. 26ATSCh. 8.10 - Prob. 27ATSCh. 8.11 - Prob. 7LTSCh. 8 - Prob. 47PP
Knowledge Booster
Similar questions
- Cembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV absorption at 245 nm, but dihydrocembrene (C20H34), the product of hydrogenation with 1 equivalent of H2, has no UV absorption. On exhaustive hydrogenation, 4 equivalents of H2 react, and octahydrocembrene, C20H40, is produced. On ozonolysis of cembrene, followed by treatment of the ozonide with zinc, four carbonylcontaining products are obtained: Propose a structure for cembrene that is consistent with its formation from geranylgeranyl diphosphate.arrow_forwardis steric hinderance not an issue here? why when reacting with KOC(CH3)3 does it yield 2-isopropyl-1-pentene and it yields 2,3-dimethyl-2hexene here?arrow_forward(ii) Draw the product R, including a detailed reaction mechanism for the conjugate addition reaction between the 1,3-dicarbonyl P and the a,ß-unsaturated ketone Q. Eto P OEt + Q OEt Et3N R (iv) Explain why conjugate addition is favoured over direct addition to Q in this case.arrow_forward
- A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2 CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under the same conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C. Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structures of A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions.arrow_forwardArrange the following groups in increasing CIP priority order: * -C-OCH3 С-ОН C–CH3 -C-NH2 I II III IV III, IV, II, I O II, I, IV, II O II, III, I, IV O I, I, IV, Iarrow_forwardDraw the organic product of the reaction below. Show stereochemistry when relevant. If the product exists as a pair of enantiomers, draw only one. Ä + T 1. LIAIH 4 2. H₂O 2 X Ś è Click and drag to start drawing a structure. 8 00. Ar ▶arrow_forward
- Consider the reaction between (1S,3S)-1-chloro-3-methylcyclopentane and methanethiol in the presence of sodium hydroxide. (a) Draw the organic product and clearly indicate stereochemistry by showing the hydrogen on the chirality centers and using wedge and dash bonds. (b) Then analyze the stereochemistry of the product. -SH C NaOH Draw only the organic product. Select /// Draw Rings More C H S Erase:arrow_forwardA 1.62 M solution of (R)-2-butanol is mixed with an equal volume of a 0.810 M solution of racemic 2-butanol, and the resulting solution is analyzed in a sample container that is 1 dm long. What observed rotation is expected? The specific rotation of (R)-2-butanol is –13.9 degrees mL g- dm-'. a = degarrow_forwardGive a rough estimate for delta H of the Pt-catalyzed hydrogenation of 1,2 dimethylcyclohexene. And draw the HOMO orbital of H2 and overlay it with the LUMO of the alkenearrow_forward
- help with #18arrow_forwardDraw a structural formula for the product formed upon hydroboration/oxidation of the alkene below. CH3 OG 1 Use wedge and hash bonds ONLY for rings. Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer. ● 981 CH3 ***** // Q Y ? [F Barrow_forward1. Predict the major product(s) for the following reactions. Indicate stereochemistry and draw enantiomers when appropriate. Show stereochemistry with two lines in the plane and a wedge and/or dash at the chirality center. No reaction mechanisms are necessary. (a) :0: (b) :0: 1. H₂O €0. Na:QH 2. H+, H₂O 1. H₂O ee.. Na:OH 2. H+, H₂O 2. (a) Copy the reaction scheme for the ester hydrolysis of methyl salicylate (see Scheme 7), make a table showing the physical properties of methyl salicylate, 3.0 M NaOH and salicylic acid, and (b) outline the experimental procedure in your laboratory notebook. Reaction NaOH(aq) ONa HCl(aq) OH ONa Methyl salicylate OH OH Salicylic acid + NaCl + -OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning