
Concept explainers
Predict When sodium metal reacts with water, it produces sodium hydroxide, hydrogen gas, and heat. Write balanced chemical equations for 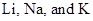 reacting with water. Use Figure 9.13 to predict the order of the amount ofheat released from least to most amount of heat released.
reacting with water. Use Figure 9.13 to predict the order of the amount ofheat released from least to most amount of heat released.
Interpretation:
The balanced chemical equations for reaction of Li, Na and K with water needs to be determined. The order of the amount of heat released from least to most needs to be determined.
Concept introduction:
The reactivity series of metals is as follows:
Lithium > Rubidium > Potassium > Calcium > Sodium > Magnesium > Aluminium > Manganese > Zinc > Iron > Nickel > Tin > Lead > Copper > Silver > Platinum > Gold
Answer to Problem 121A
Lithium reaction releases maximum amount of heat which is followed by the potassium reaction and lastly by the sodium reaction.
Following are the complete balanced reactions:
Explanation of Solution
The reactivity series of metals is as follows:
Lithium > Rubidium > Potassium > Calcium > Sodium > Magnesium > Aluminium > Manganese > Zinc > Iron > Nickel > Tin > Lead > Copper > Silver > Platinum > Gold
On the basis of the order of reactivity series of metals we can conclude that Lithium reaction releases maximum amount of heat which is followed by the potassium reaction and lastly by the sodium reaction.
Following are the complete balanced reactions:
Lithium reaction releases maximum amount of heat which is followed by the potassium reaction and lastly by the sodium reaction. This can be concluded on the basis of reactivity series of metals.
Following are the complete balanced reactions:
Chapter 9 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Inorganic Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: Structure and Properties (2nd Edition)
CHEMISTRY-TEXT
Organic Chemistry (9th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





