
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
6th Edition
ISBN: 9780135765982
Author: Karen Timberlake, William Timberlake
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 48UTC
The chapter sections lo review are shown in parentheses at the end of each problem
If green spheres represent chlorine atoms, yellow-green spheres represent fluorine atoms, white spheres represent hydrogen atoms, and all the molecules are gases. (9.2. 9.4)
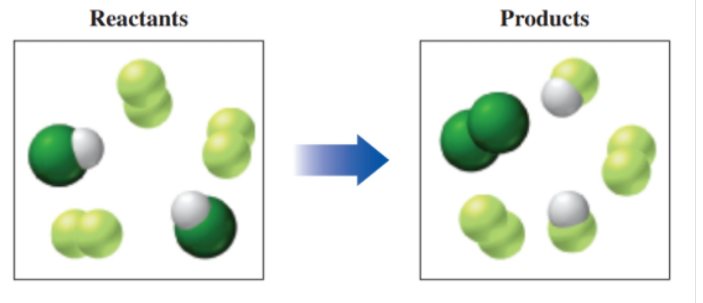
a. write a balanced equation for the reaction.
b. identify the limiting reactant.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(6.3)The volume of a gas is 0.953 L at 30.0 °C. If the gas is heated to 60.0 °C, what would be the volume of the gas (in L) at this
temperature?
O 1.05 L
O 0.867 L
O 1.91 L
O 0.477 L
(5.8)Which of the following reactions will form a gaseous product?
O H₂CO3(aq) + Pb(NO3)2(aq)
O NaOH(aq) + HNO3(aq)
O None of these
O Na₂SO3(aq) + H₂SO4(aq)
◄ Previous
(3.10)A compound has a molar mass of 126.12 g/mol and its empirical formula is C₂H₂O.
Find the molecular formula and fill in each blank with a whole number.
H
< Previous
Chapter 9 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
Ch. 9.1 - Calculate the total mass of the reactants and the...Ch. 9.1 - Calculate the total mass of the reactants and the...Ch. 9.2 - Calculate the total mass of the reactants and the...Ch. 9.2 - Write all of the mole-mole factors for each of the...Ch. 9.2 - For the chemical equations in problem 9.3, write...Ch. 9.2 - For the chemical equations in problem 9.4, write...Ch. 9.2 - The chemical reaction of hydrogen with oxygen...Ch. 9.2 - Ammonia is produced by the chemical reaction of...Ch. 9.2 - Carbon disulfide and carbon monoxide are produced...Ch. 9.2 - In the acetylene torch, acetylene gas burns in...
Ch. 9.3 - Sodium reacts with oxygen to produce sodium oxide....Ch. 9.3 - Nitrogen reacts with hydrogen to produce ammonia....Ch. 9.3 - Ammonia and oxygen react to form nitrogen and...Ch. 9.3 - Iron(III) oxide reacts with carbon to give iron...Ch. 9.3 - Prob. 15PPCh. 9.3 - Calcium cyanamide, CaCN2 , reacts with water to...Ch. 9.3 - Prob. 17PPCh. 9.3 - Prob. 18PPCh. 9.4 - A taxi company has 10 taxis. a. On a certain day,...Ch. 9.4 - Prob. 20PPCh. 9.4 - Prob. 21PPCh. 9.4 - Iron and oxygen react to form iron(III) oxide....Ch. 9.4 - For each of the following reactions, 20.0 g of...Ch. 9.4 - For each of the following reactions, 20.0 g of...Ch. 9.4 - For each of the following reactions, calculate the...Ch. 9.4 - For each of the following reactions, calculate the...Ch. 9.5 - Carbon disulfide is produced by the reaction of...Ch. 9.5 - Iron (III) oxide reacts with carbon monoxide to...Ch. 9.5 - Aluminum reacts with oxygen to produce aluminum...Ch. 9.5 - Propane ( C3H8 ) bums in oxygen to produce carbon...Ch. 9.5 - Prob. 31PPCh. 9.5 - When 56.6 g of calcium is reacted with nitrogen...Ch. 9.6 - In an exothermic reaction, is the energy of the...Ch. 9.6 - In an endothermic reaction, is the energy of the...Ch. 9.6 - Prob. 35PPCh. 9.6 - Prob. 36PPCh. 9.6 - Classify each of the following as exothermic or...Ch. 9.6 - Classify each of the following as exothermic or...Ch. 9.6 - Prob. 39PPCh. 9.6 - a. How many kilojoules are released when 75.0 g of...Ch. 9.6 - Prob. 41PPCh. 9.6 - Calculate the energy change for the reaction...Ch. 9.6 - Prob. 43PPCh. 9.6 - Prob. 44PPCh. 9.6 - In one step in the synthesis of the insecticide...Ch. 9.6 - Another widely used insecticide is carbofuran...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - The chapter sections lo review are shown in...Ch. 9 - Prob. 55APPCh. 9 - Prob. 56APPCh. 9 - Prob. 57APPCh. 9 - Prob. 58APPCh. 9 - Prob. 59APPCh. 9 - Prob. 60APPCh. 9 - Pentane gas ( C5H12 ) reacts with oxygen to...Ch. 9 - Prob. 62APPCh. 9 - Prob. 63APPCh. 9 - Prob. 64APPCh. 9 - Prob. 65APPCh. 9 - Prob. 66APPCh. 9 - Prob. 67APPCh. 9 - Prob. 68APPCh. 9 - Prob. 69APPCh. 9 - The equation for the reaction of iron and oxygen...Ch. 9 - Prob. 71APPCh. 9 - Prob. 72APPCh. 9 - Prob. 73CPCh. 9 - Prob. 74CPCh. 9 - Prob. 75CPCh. 9 - The following problems are related to the topics...Ch. 9 - Prob. 77CPCh. 9 - Prob. 78CPCh. 9 - Prob. 79CPCh. 9 - Prob. 80CPCh. 9 - The following problems are related to the topics...Ch. 9 - Prob. 82CPCh. 9 - Prob. 83CPCh. 9 - The following problems are related to the topics...Ch. 9 - Prob. 85CPCh. 9 - Prob. 86CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 9. Which element is oxidized in the following reaction: 2FECI2 + Cl2 → 2FECI3 if Fe goes from +2 to +3 and CI goes from 0 to –2? Fe Neither element CI Cl and Fe GET IT NOW New version available! (3.0.220) PREVIOUSarrow_forward9.116 Lithium chloride has a solubility of 55 g of LiCl in 100. g of H,O at 25 °C. Determine if each of the following mixtures forms an unsaturated or saturated solution at 25 °C: (9.3) a. adding 10 g of LiCl to 15 g of H;O b. adding 25 g of LİCI to 50. g of H,0 c. adding 75 g of LiCl to 150. g of H,O ne on nearrow_forwardBarium Hydroxide has a solubility of 4.68 g Ba(OH)2 in 100. g H2O at 25°C. Determine if each of the following solutions will be saturated or unsaturated at 25°C: (9.3) a) adding 32 g of Ba(OH)2 to 990 g of H2O b) adding 7.0 g of Ba(OH)2 to 125 g of H2O c) adding 22 g of Ba(OH)2 to 350. g of H2Oarrow_forward
- 9.2 An unknown salt is found to contain 30,6% of water. The formula mass for anhydrous salt (AS) is 100 amu. 9.2.1 Calculate the amount of water bonded with an unknown salt. 9.2.2 An anhydrous salt above consists of a metal with a molar mass of 40. (a) Write down the chemical symbol of this metal. (b) To which group does the metal belongs? 9.2.3 Write down the chemical formula of an anhydrous salt above.arrow_forward5. Which element is reduced in the following reaction: 2Mg + O2 → 2M9O if Mg goes from 0 to +2 and O goes from 0 to -2? O Mg and O Neither element Mg New version available! (3.0.220) GET IT NOW PRarrow_forwardH₂O₂ + KMnO4 + H₂SO4 O₂ + MnSO4 + K₂SO4+H₂O 1. How many moles of the oxidized product is formed in the reaction? (2,1,3,4) 2. How many electrons does the reducing agent loss?(5,3,7,1)arrow_forward
- (5.2, Similar to For More Practice 5.1) What mass of NaOH (in grams) do you need to make 250.0 mL of a 1.50 M NaOH solution? O 6.67 g O 22.3 g O 44.6 g O 15.0 garrow_forwardHonors Chemistry-4th Hour- Dr. Paul / Gases / Lesson 148 11. Which state of matter is most accurately described by the kinetic molecular theory? O gas aqueous solid O liquid available! (3.0.227)arrow_forwardCalculate the mass, in grams, for each of the following using Table 3.7: (3.5)a. a sample of silver (Ag) that absorbs 1650 J when its temperature increases from 65 °C to 187 °Cb. an iron (Fe) bar that loses 2.52 kJ when its temperature decreases from 252 °C to 75 °Carrow_forward
- 6.149 Indicate the major type of intermolecular forces-(1) ionic bonds, (2) dipole-dipole attractions, (3) hydrogen bonds, (4) dispersion forces-that occurs between particles of the following: (6.9) a. NF3 d. Cs,0 с. Brz f. CH;OH b. CIF e. C4H10arrow_forwardIn an experiment, 7.84 g of sulphuric acid reacts with potassium carbonate. H2SO4 + K2CO3 —> K2SO4 + H2O + CO2 Calculate: the mass of potassium carbonate consumed (11.0g)arrow_forwardYou have a large bottle of 10.0 M hydrochloric acid and you want to make 6.0 L of 3.0 M hydrochloric acid. How much concentrated do you need and how much water? (1.8, 4.2)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY