
Interpretation:
Balance the equation depicting the total combustion of methanol which results in the formation of carbon dioxide and water and calculate the mass of carbon dioxide formed after the compete combustion of 775 g of methanol.
Concept Introduction:
Methanol is the simplest form of alcohol. Methyl alcohol, wood alcohol and wood spirit are some of the names of methanol. Its chemical formula is 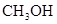 It is a methyl group which is linked to a hydroxyl group. It is abbreviated as MeOH. Earlier it was produced by the destructive distillation of woods but now-a-days it is prepared by the combination of hydrogen and carbon monoxide gas with the help of a catalyst.
It is a methyl group which is linked to a hydroxyl group. It is abbreviated as MeOH. Earlier it was produced by the destructive distillation of woods but now-a-days it is prepared by the combination of hydrogen and carbon monoxide gas with the help of a catalyst.
Some of its features are mentioned below:
- Light
- Volatile
- Colorless
- Flammable liquid
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Chemistry For Changing Times (14th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





