
Label each ether and alcohol in brevenal, a marine natural product. Classify each alcohol as
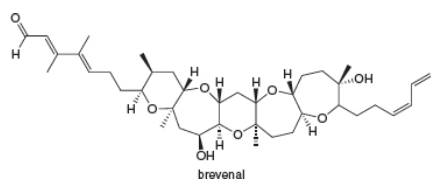
Interpretation: Each ether and alcohol in brevenal is to be labeled and each alcohol is to be classified as
Concept introduction: Alcohols and ethers are the functional groups that involve
Answer to Problem 9.1P
Each ether and alcohol in brevenal is labeled as shown below.
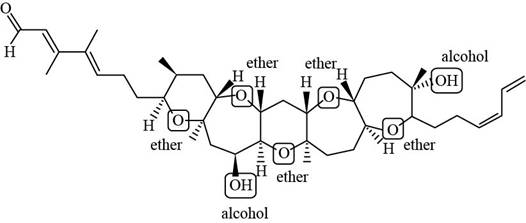
Explanation of Solution
The structure of brevenal (given compound) is,
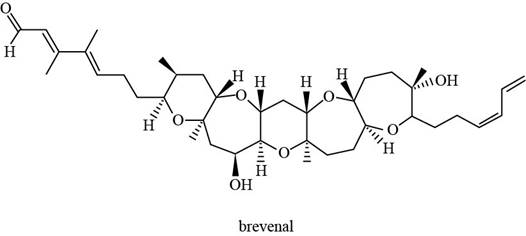
Figure 1
Alcohols and ethers are the functional groups that involve
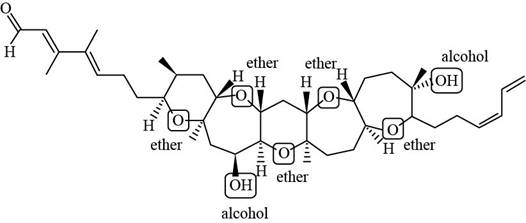
Figure 2
Alcohols are classified as
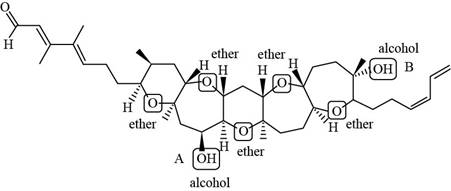
Figure 3
In Figure 3, the carbon atom of alcohol (A) is bonded to two
If the number of carbon atom bonded to the carbon atom with the hydroxyl group is one, then it is classified as
Thus, each alcohol in brevenal is classified as,
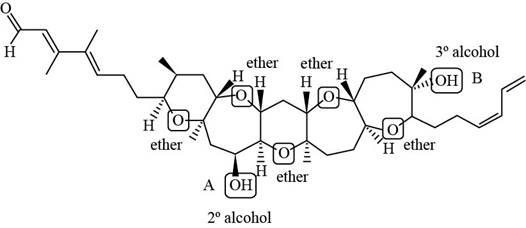
Figure 4
Each ether and alcohol in brevenal is labeled in Figure 2.
Want to see more full solutions like this?
Chapter 9 Solutions
ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
Organic Chemistry - Standalone book
Chemistry
Chemistry: Matter and Change
Principles of Chemistry: A Molecular Approach (3rd Edition)
- Name the following alcohols utilizing the IUPAC nomenclature rules a. CH3 CH3-CH,-C–CH2–CH,–CH3 ОН b. CH; —CH, —СН— СН, — СН; CH —ОН с. CH3 CH3- -ОНarrow_forwardOrganic Reaction Write an equation for the oxidation of each alcohol. Use [O] above the arrow to indicate an oxidizing agent. If no reaction occurs, write "no reaction" after the arrow. 1. CH3CH2CH2CH2CH2OH CH₂ CH₂CCH₂CH₂ ОН CH CHCH CH CH CH, ОНarrow_forwardH H H 11 The compound H–Ç–C-C- 0–c-H is an example of an |1 H HH H. ether. O True O Falsearrow_forward
- Define Preparation of Ethers ?arrow_forwardWhich would be a secondary alcohol? . a) CH3 – CH2 – CH2OH b) CH3 – CH – CH3 OH c) CH3 – CH – CH2 OH OH d) OH CH3 – C – CH3 CH3 Oarrow_forwardFor each of the following, draw a structural formula that meets the stated requirements, then name each structureyour drew (unless otherwise indicated) according to either IUPAC or common nomenclature.arrow_forward
- Which of the following is a secondary alcohol? O A. но CH3 CH2CH3 В. CH2CH2CH3 CH3 CH2-C-CH2CH3 Он O C. CH3 CH 2-CH-CH3 Он D. CH3CH2-CH-CH2CH2CH3 ČH2CH2CH2OHarrow_forwardDetermine whether the compound is a primary, secondary, or tertiary alcohol.arrow_forwardIdentify each compound as an ether, hemiacetal, or acetal.arrow_forward
- в. Odoriferous Organic Compounds Below each company identify the source or use as recognized by familiar odor. CH3 c-CH-CH,CH,c=CHCH,OH CH3 CH, C-CH-CH,CH,-C-CH-C CH3 CH CH3 H. Geraniol Citral CH 3 С -Н CH3 CH3- CH OCH3 он CH3 CH3 Pinene Carvone Vanillin CH он CH OCH3 CHC-CH ČH,CH-CH2 CH CH3 CH, Menthol Camphor Eugenol C1 co,CH, OH CH=CH-C Methyl salicylate Cinnamaldehyde p-Dichlorobenzenearrow_forwardGive the common name for NN OH T CH3-CH₂-C-N-CH₂-CH3 Spell out the common name of the compound. Give the IUPAC name for 0 CH₂ || CH₂-C-N-CH₂-CH₂-CH₂, Spell out the IUPAC name of the compound. diethylamine Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. HE CONT HOarrow_forward1: Give at least five (5) uses of Etherin medicine. CH3 CH,-0-C-CH, CH, common name: methyl-t-butyl ether (MTBE) IUPAC name: 2-methoxy-2-methylpropane diphenyl ether or phenyl ether phenoxybenzene Analyze the given ether. Then describe the structural formula.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





