
What
results.
a. 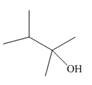 b.
b. 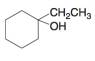 c.
c. 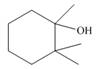 d.
d.
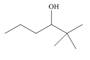
(a)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
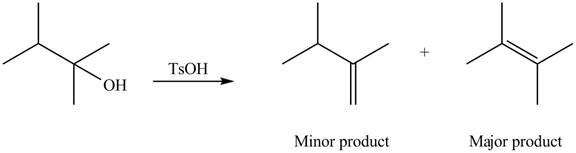
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with
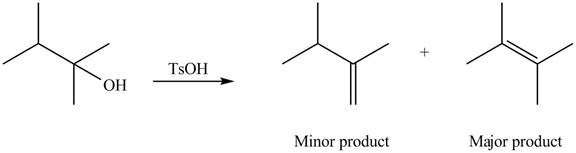
Figure 1
The alkenes formed by the treatment of given alcohol with
(b)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with

Figure 2
The alkenes formed by the treatment of given alcohol with
(c)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
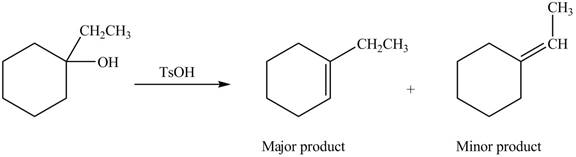
Explanation of Solution
The given alcohol contains three
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with
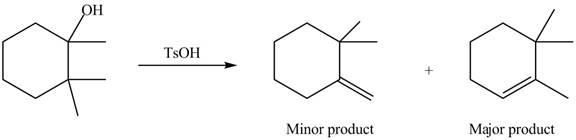
Figure 3
The alkenes formed by the treatment of given alcohol with
(d)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkene formed by the treatment of given alcohol with
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
The alkenes formed by the treatment of given alcohol with
(e)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
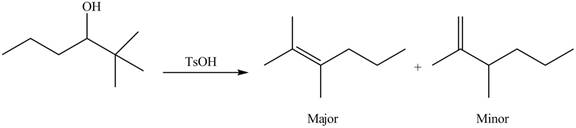
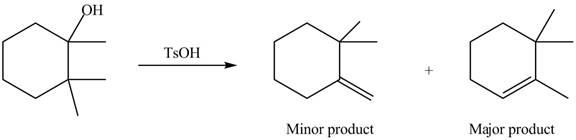
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with

Figure 4
The alkenes formed by the treatment of given alcohol with
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry -Study Guide / Solution Manual (Custom)
- ALCOHOLS 1. WHY IS ETHANOL MORE SOLUBLE IN WATER THAN 1-HEXANOL? 2. WHAT IS DENATURED ALCOHOL? AND WHY IS ALCOHOL DENATURED? ETHER 1. WHY DOES DIETHYL ETHER HAVE MUCH LOWER BOILING POINT THAN 1-BUTANOL?arrow_forwardExplain Addition of Alcohols—Acetal Formation ?arrow_forwardDraw the product formed when (CH3)2CHOH is treated with each reagent (d, e and f)arrow_forward
- Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. c. CH3CH2Cl, AlCl3 l. product in (c), then KMnO4arrow_forwardAnswer each question using the ball-and-stick model of compound A. Draw a constitutional isomer that contains an ether and give its IUPACname. Draw the products formed (including stereochemistry) when A istreated with each reagent: [1] NaH; [2] H2SO4; [3] POCl3, pyridine; [4]HCl; [5] SOCl2, pyridine; [6] TsCl, pyridinearrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forward
- Draw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.arrow_forwardWhat ester and Grignard reagent are needed to prepare each alcohol?arrow_forwardMolecule Type Boiling point (°C) CH3CH2CH3 Alkane -42 CH3CHO Aldehyde +21 CH3CH2OH Alcohol +78 i. Why is the boiling point of the aldehyde greater than that of the alkane?ii. Why is the boiling point of alcohol the highest?iii. Explain why the solubility of aldehydes and alcohols falls as the molecules get bigger.arrow_forward
- (a) Draw the products formed when propane is heated with Br2. (b) Label major and minor products and explain with an energy diagram.arrow_forward(a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: [1] NaBH4, CH3OH; [2] CH3MgBr, then H2O; [3] Ph3P = CHOCH3; [4] CH3CH2CH2NH2, mild acid; [5] HOCH2CH2CH2OH, H+.arrow_forwardn-Butyl methyl ether is an isomer of MTBE and has a boiling point of 70 oC. Explain why the boiling point is significantly different compared to MTBE.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
