
Concept explainers
Write Lewis dot symbols for the following atoms and ions: (a) I, (b) I−, (c) S, (d) S2−, (e) P, (f) P3−, (g) Na, (h) Na+, (i) Mg, (j) Mg2+, (k) Al, (l) Al3+, (m) Pb, (n) Pb2+.
(a)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
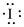
(b)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
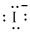
(c)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
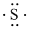
(d)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
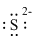
(e)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
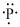
(f)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
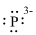
(g)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
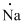
(h)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
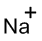
(i)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
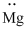
(j)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
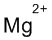
(k)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
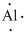
(l)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
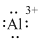
(m)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
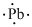
(n)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
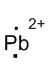
Want to see more full solutions like this?
Chapter 9 Solutions
CHEMISTRY STUD.SOL.MANUAL >C<
- Write a Lewis symbol for atom or ion. Mg2+arrow_forwardWhich of the following ionic compounds would be expected to have the highest lattice energy? RbBr, KF, NaI, MgBr2arrow_forwardWhich of the following atoms would be expected to form negative ions in binary ionic compounds and which would be expected to form positive ions: P, I, Mg, Cl, In, Cs, O, Pb, Co? Which of the following atoms would be expected to form negative ions in binary ionic compounds and which would be expected to form positive ions: Br, Ca, Na, N, F, Al, Sn, S, Cd?arrow_forward
- Which of the following atoms would be expected to form negative ions in binary ionic compounds and which would be expected to form positive ions: Br, Ca, Na, N, F, Al, Sn, S, Cd?arrow_forwardWrite Lewis dot symbols for the following atoms and ions. (a) I (b) I− (c) S (d) S2−arrow_forwardWrite Lewis structures for the following: (c) C2F6 (contains a C¬C bond), (d) AsO3 3 -, (e) H2SO3 (H is bonded to O), (f) NH2Cl.. Arrange the bonds in each of the following sets in order of increasing polarity: (a) C¬F, O¬F, Be¬F; (b) O¬Cl, S¬Br, C¬P; (c) C¬S, B¬F, N¬O. What is the Lewis symbol for each of the following atoms or ions? (a) K, (b) As, (c) Sn2 + , (d) N3 Write electron configurations for the following ions and determine which have noble-gas configurations: (a) Cd2+, (b) P3-, (c) Zr4+arrow_forward
- Write the Lewis symbols of the ions in each of the following ionic compounds and the Lewis symbols of the atom from which they are formed:(a) MgS(b) Al2O3(c) GaCl3(d) K2O(e) Li3N(f) KFarrow_forwardwe would predict that the imaginary ionic compound XF would have a (higher /lower) lattice energy than YyF2 becausearrow_forwardWrite a Lewis symbol for atom or ion. Mgarrow_forward
- Write the Lewis symbols that represent the ions in ionic compound. SrBr2arrow_forwardWrite the Lewis symbols for Ca2+ and Cl-arrow_forwardWrite all resonance structures of chlorobenzene, C6H5Cl, a molecule with the same cyclic structure as benzene. In all structures, keep the CCl bond as a single bond. Which resonance structures are the most important?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





