
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 9.3, Problem 9.20P
Interpretation Introduction
Interpretation:
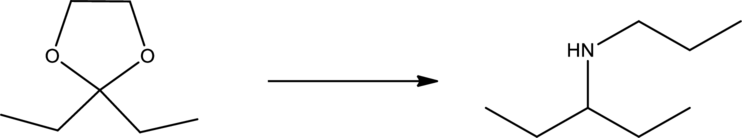
Efficient synthesis for the given transformation has to be suggested.
Concept Introduction:
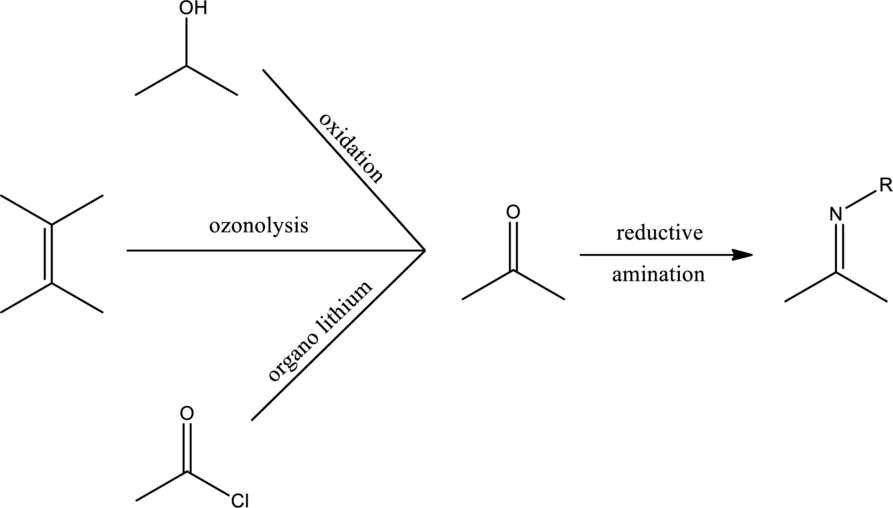
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose syntheses for the product shown on the left starting from the indicated compound (shown on the right). Your answer should include a retrosynthetic analysis and a proposed synthetic route with reagents.
Propose an efficient synthesis for the following transformation. Hint: For the first problem you will need to use the ozonolysis reaction.
Provide an efficient synthesis for each of the following transformations. Include your
retrosynthetic analysis, showing products for each step of the reaction sequence. You are not required
to write a mechanism for any of these.
Chapter 9 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...
Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Prob. 9.18PCh. 9.3 - Prob. 9.19PCh. 9.3 - Prob. 9.20PCh. 9.3 - Prob. 9.21PCh. 9.3 - Prob. 9.22PCh. 9.4 - Prob. 9.24PCh. 9.4 - Prob. 9.25PCh. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...
Knowledge Booster
Similar questions
- Show how you would accomplish the following synthetic transformations. You may useany necessary reagents. ) ethyl benzoate S N-ethylbenzamidearrow_forwardShow how you could accomplish the following transformation. Be sure to show the structures of the products after each reaction you propose as well as the reagents required for each reaction. You do not need to show reaction mechanisms or a retrosynthesis.arrow_forwardHow would you carry out the following transformation? Tell what reagents you would use in each case.arrow_forward
- Propose a multi-step synthesis of the target molecule shown at the right, using the starting materials on the left and any other reagents you need. Show the reagents needed for each step and the product of each step. You will need 4 reaction arrows.arrow_forwardShow how you would accomplish the following syntheses.benzene ¡ p@methoxybenzaldehydearrow_forwardShow how to synthesize the following compound using either the malonic ester synthesis or the acetoacetic ester synthesis. Q.) Cyclopropanecarboxylic acidarrow_forward
- Show how you would perform the following transformation using readily available reagents:arrow_forwardShow what reagents are necessary to make the following acetal from a ketone. Themechanism is not necessary.arrow_forwardHow would you synthesize the methyl ketone shown below via the acetoacetic ester synthesis? You must show the reactions/reagents used in their correct orderarrow_forward
- If methanol rather than water is added at the end of a Hell–Volhard–Zelinskii reaction, an ester rather than an acid is produced. Show how you would carry out the following transformation, and propose a mechanism for the ester forming step.arrow_forwardFor the following reaction schemes, fill in the appropriate reagents and starting materials you would use to transform the acetoacetic ester and acetone into nabumetone, an NSAID drug used for arthritis and joint pain. Write the correct reagents/reactants next to the number of steps for each transformation.arrow_forwardPropose a synthetic route to carry out the following Transformations.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
