
Concept explainers
(a)
Interpretation:
The major elimination product obtained when the given alcohol is heated in the presence of sulphuric acid has to be has to be drawn.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction of alcohol with strong acid like sulfuric acid is known as dehydration reaction.
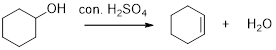
E1 dehydration reaction of secondary and tertiary alcohols:
The alcohols react with acids like hydrochloric acid or hydrobromic which yield the corresponding stable carbocation intermediate. The elimination of hydrogen from the beta carbon results in the
Carbocation: It is carbon ion that bears a positive charge on it.
The increasing stability order of carbocation is as follows,
Primary carbocation < secondary carbocation < tertiary carbocation
(b)
Interpretation:
The major elimination product obtained when the given alcohol is heated in the presence of sulphuric acid has to be has to be drawn.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction of alcohol with strong acid like sulfuric acid is known as dehydration reaction.
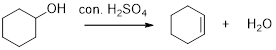
E1 dehydration reaction of secondary and tertiary alcohols:
The alcohols react with acids like hydrochloric acid or hydrobromic which yield the corresponding stable carbocation intermediate. The elimination of hydrogen from the beta carbon results in the alkene product. Thus the removal of water molecule occurs in the dehydration process and the major product of the acid-catalysed dehydration reaction will be the more substituted product.
Carbocation: It is carbon ion that bears a positive charge on it.
The increasing stability order of carbocation is as follows,
Primary carbocation < secondary carbocation < tertiary carbocation
(c)
Interpretation:
The major elimination product obtained when the given alcohol is heated in the presence of sulphuric acid has to be has to be drawn.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction of alcohol with strong acid like sulfuric acid is known as dehydration reaction.
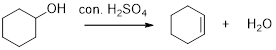
E2 dehydration of primary alcohols:
Due to the unstability of the primary carbocation, the dehydration of primary alcohol is an E2 reaction.
In the E2 reaction, protonation of the most basic atom occurs and then base will remove a proton from the beta carbon.
(d)
Interpretation:
The major elimination product obtained when the given alcohol is heated in the presence of sulphuric acid has to be has to be drawn.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction of alcohol with strong acid like sulfuric acid is known as dehydration reaction.
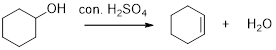
E1 dehydration reaction of secondary and tertiary alcohols:
The alcohols react with acids like hydrochloric acid or hydrobromic which yield the corresponding stable carbocation intermediate. The elimination of hydrogen from the beta carbon results in the alkene product. Thus the removal of water molecule occurs in the dehydration process and the major product of the acid-catalysed dehydration reaction will be the more substituted product.
Carbocation: It is carbon ion that bears a positive charge on it.
The increasing stability order of carbocation is as follows,
Primary carbocation < secondary carbocation < tertiary carbocation
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Essential Organic Chemistry (3rd Edition)
- draw the structure of the expected product when the compound provided undergoes Claisen rearrangement.Include isomerization to a phenol IF appropriate.arrow_forwardDraw the major product(s) obtained from the acid-catalyzed hydration of the following alkene: CH3CH2CH2CH=CH2arrow_forwardWhat are the major products obtained when each of the following ethers is heated with one equivalent of HI?arrow_forward
- What product is formed when 3-methyl-2-cyclohexenone reacts with each of the following reagents? a. CH3MgBr followed by H3O+ b. (CH3CH2)2CuLi followed by H3O+ c. HBr d. CH3CH2SHarrow_forwardRank the compounds in each group in order of increasing reactivity in nucleophilic acyl substitution. CH3CH2CO2H, (CH3CH2CO)2O, CH3CH2CONHCH3arrow_forwardDraw the favored product(s) of the following reactions (organic chemistry)arrow_forward
- What products are obtained from the reaction of the following compound with H2CrO4 + Δ?arrow_forwardDraw the major elimination product for the reaction of 2-bromo2-methylbutane with A) KOH B) KOtBu C) 1-butanol D) 1-propanolarrow_forwardWhat product is obtained when the following compound undergoes two successive elimination reactions?arrow_forward
- Be sure to answer all parts. Draw the product formed when the following alkene is treated with either [1] HBr alone and [2] HBr in the presence of peroxides. CH2CHCH2CH2CH2CH3arrow_forwardM8. draw the products formed in the following transformations A-Carrow_forwardDraw the most stable enol form for each of the following keto tautomersarrow_forward
