
Concept explainers
Practice Problem ATTEMPT
ATTEMPT
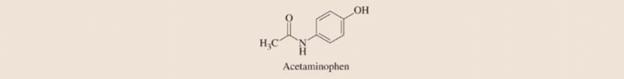
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
CHEMISTRY W/CONNECT ACCESS CODE (LL)
Additional Science Textbook Solutions
Organic Chemistry
Fundamentals of Heat and Mass Transfer
General Chemistry: Atoms First
Chemistry: Atoms First
Organic Chemistry
Organic Chemistry (8th Edition)
- What would be the shape of a molecule where the central atom has two nonbonding pairs while also covalently bonded to two other atoms? Group of answer choices bent pyramidal tetrahedral linear T-shapedarrow_forwardPotential energy diagram for Qr2 molecule. What is the bond energy in kJ/mol in the Qr-Qr bond? In which region of the curve are the repulsive forces stronger than the attractive forces? In which region of the curve are the repulsive and attractive forces the same?arrow_forwardAnswer the following question about acetonitrile (CH3C≡N:). Question: Label all bonds as polar or nonpolar ?arrow_forward
- Which of the following is/are polar bond(s) (choose all that apply)? Group of answer choices C-Cl C-S C-H Cl-Cl C-Carrow_forwardWhich of the following statements can be correctly applied to polar molecules? Select all that apply. I chose C & Darrow_forwardWhich of the molecules, CO2, H2O, NH3, and BF3, will be polar? Question 18 options: a) CO2, NH3 and BF3 b) CO2, H2O and NH3 c) H2O and NH3 d) CO2 and BF3 e) H2O and BF3arrow_forward
- T1 Choose correct answer: Find the molecule that does not octet principle. A) CI4 B) PCI5 C) PCI3 D) CF4arrow_forwardDo the same as above for the formula C6H10. Draw three isomers such that each atom makes the appropriate number of bonds, as indicated by their Lewis Dot Symbol. (There are many structures that work! Hint: These molecules will feature C-C pi bonds…)arrow_forwardQuestion:Theoretical Question: The hybridization of an atom in a molecule determines its geometry and reactivity. Consider the molecule X₃Y₂, where X represents an atom with sp² hybridization and Y represents an atom with sp³ hybridization. How would the hybridization and geometry of the central atom in X₃Y₂ affect its reactivity? Provide a detailed explanation.arrow_forward
- Statement 1: Compound1 is more stable since it has lowertorsion al energy Statement 2: It is why its lineargeometry resists the rotation of atoms in its structurearrow_forwardA) Which of the following bonds are nonpolar? Choose all that apply Choices: O=O double bond C=O double bond C-O single bond C=C double bond C-H single bond N-H single bond O-H single bond H-H single bond B) Which of the following bonds are polar covalent? Choices: N-H single bond C=O double bond C-O single bond C-H single bond C-C single bond N-F single bond S-H single bond O=O double bond Thank you so Much!!arrow_forwardDraw a molecule that has a trigonal pyramidal geometry using D as the central atom and G as the outer atoms picture does not have to show 3 dimensions and outer atoms do not need ione pairs. G is more electronegative than D State wether this molecule is polar or non polar? What are it's bond angles?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





