
Concept explainers
a)
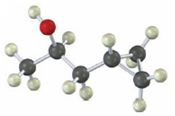
Interpretation:
Starting from any compound having four or less number of carbon atoms how the compound shown can be prepared is to be stated.
Concept introduction:
A cyclopropane ring is formed in Simmons-Smith reaction when an alkene is treated with CH2Cl2/Zn-Cu couple. Higher
To state:
Starting from any compound having four or less number of carbon atoms how the compound shown can be prepared.
b)
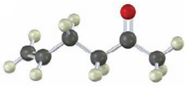
Interpretation:
Starting from any compound having four or less number of carbon atoms how the compound shown can be prepared is to be stated.
Concept introduction:
The compound shown has a keto group at C2 and a double bond between C5 & C6. The keto group can be introduced by hydration of an alkyne with aqueous H2SO4 in the presence of HgSO4. An enol obtained during hydration upon tautomerization yields the
To state:
Starting from any compound having four or less number of carbon atoms how the compound shown can be prepared.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- Show how the following compound can be prepared from starting materials that have no more than five carbons:arrow_forwardHow do you synthesize 3-oxocyclohexanecarboxylic acid from 2-cyclohexenone?give earrow_forwardUsing acetonitrile (CH3CN) and CO2 as your only sources of carbon atoms, identify how you could prepare each of the following compounds:arrow_forward
- Show how lidocaine, one of the most widely used injectable anesthetics, can be prepared from benzene and compounds containing no more than four carbonsarrow_forward2. How would you prepare the following substances from 2-cyclohexenone? More than onestep may be needed.(c) 3-Oxocyclohenanecarboxylic acidarrow_forwardShow how you would synthesize the following compounds, starting with acetylene and any compounds containing nomore than four carbon atoms. pentanal, CH3CH2CH2CH2CHOarrow_forward
- Starting from cyclohexanol, show how to prepare: Show all reagents and molecules synthesized along the way after each step. You do not have to use curved arrows.arrow_forwardHow would you prepare the following carboxylic acids?arrow_forwardUsing an alkyl halide and a thiol as starting materials, how would you prepare the following thioethers?arrow_forward
