
Concept explainers
a)
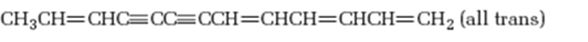
Interpretation:
The hydrocarbon shown is isolated from sun flower family. It is to be named following the IUPAC rules.
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is assigned to it.
To name:
The hydrocarbon shown, isolated from sun flower family, according to the IUPAC rules.
b)
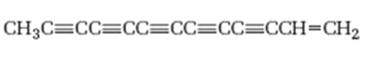
Interpretation:
The hydrocarbon shown is isolated from sun flower family. It is to be named following the IUPAC rules.
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is assigned to it.
To name:
The hydrocarbon shown, isolated from sun flower family, according to the IUPAC rules.
Trending nowThis is a popular solution!

Chapter 9 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forwardWhat reactions and reagents can be used to make phenol from benzene if electrophilic aromatic substitution reactions are excluded and benzene is the only source of carbon?arrow_forwardA 6-membered heterocycle aromatic ring with two N atoms positioned two carbons apart. a Pyridine b Pyrazine c Purine d Pyrrolearrow_forward
- When a substituted benzene undergoes an electrophilic aromatic substitution reaction, where doesthe new substituent attach itself?arrow_forwardWhich reagents and solvents can be used to synthesize (R)-2-cyanopentane from (R)-2-chloropentane?arrow_forwardComplete the following syntheses – they may be two- or three-step processes. Include any necessary catalysts or reaction conditions. a) Prepare propanone from 1-propanolarrow_forward
- 13arrow_forwardComplete the following syntheses – they may be two- or three-step processes. Include any necessary catalysts or reaction conditions. b) Prepare ethanoic acid from ethenearrow_forwardTrue or False Considering that two carbon chains have equal number of carbons, but one has Fluorine and the other has Iodine, the one with iodine will have a higher boiling point. Mild oxidation of alkenes results to similar product as that of nucleophilic addition of water to aldehydes.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

