
Concept explainers
a)
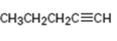
Interpretation:
How to synthesize 1-pentyne from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal
To state:
How to synthesize 1-pentyne from acetylene using any alkyl halide with four or fewer number of carbons.
b)
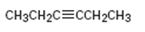
Interpretation:
How to synthesize 3-hexyne from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkyne needed. Acetylene has two acidic hydrogens. Both hydrogens can be replaced by alkyl groups through this process.
To state:
How to synthesize 3-hexyne from acetylene using any alkyl halide with four or fewer number of carbons.
c)
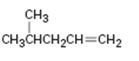
Interpretation:
How to synthesize 4-methyl-1-pentene from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkyne needed. The alkyne can be reduced to the corresponding
To state:
How to synthesize 4-methyl-1-pentene from acetylene using any alkyl halide with four or fewer number of carbons.
d)
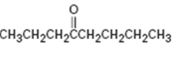
Interpretation:
How to synthesize 4-octanone from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkyne needed. The alkyne undergoes hydration when treated with dilute H3SO4 in the presence of HgSO4 to yield an enol which tautomerizes to a
To state:
How to synthesize 4-octanone from acetylene using any alkyl halide with four or fewer number of carbons.
e)
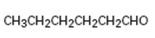
Interpretation:
How to synthesize hexanal from acetylene using any alkyl halide with four or fewer number of carbons is to be shown.
Concept introduction:
Terminal alkynes can be converted into their alkynides by treating with NaNH3 in liquid NH3. The alkynides when treated with alkyl halides with the required number of carbons yield the higher alkynes needed. The alkynes yield enols with OH on terminal carbon in hydroboration-oxidation reaction which tautomerize to yield
To state:
How to synthesize hexanal from acetylene using any alkyl halide with four or fewer number of carbons.
Trending nowThis is a popular solution!

Chapter 9 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents. cyclohexanol and BF3arrow_forward4. Synthesize the following compound. You may use benzene, acetylene, ethanol and any inorganic reagents. O₂Narrow_forwardHow to synthesize 1-methylcyclohexene from cyclohexanone?arrow_forward
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with CH3CH2COCl, AlCl3arrow_forwardHow would you convert ethanoic acid into benzene?arrow_forwardShow how lidocaine, one of the most widely used injectable anesthetics, can be prepared from benzene and compounds containing no more than fourcarbons.arrow_forward
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents.(a) HNO3, H2SO4arrow_forwardHow do you synthesize a cyclohexane-1,2,3-triol from a cyclohexanearrow_forwardStarting with bromocyclohexane, how can each of the following compounds be prepared?arrow_forward
- VShow how you would convert 2-methylcyclopentanol to the following products. Any of these products may be used as thereactant in any subsequent part of this problem.(a) 1-methylcyclopentenearrow_forwardShow how the compound can be prepared from acetylene and alkyl halides with four or fewer carbon atoms. CH3 H;Carrow_forwardhow can I synthesize 2,3,4-trimethylcyclopentadiene starting from a benzene ring or an organic compond with 3 carbons or less?arrow_forward
