
a)
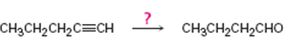
Interpretation:
How to synthesize butanal from 1-pentyne is to be shown.
Concept introduction:
To show:
How to synthesize butanal from 1-pentyne.
b)
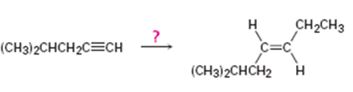
Interpretation:
How to synthesize trans- 6-methyl-3 heptene from 4-methyl-1-pentyne is to be shown.
Concept introduction:
Higher alkynes can be prepared first by converting the lower alkyne in to an alkynide by reacting with NaNH2 in liquid NH3 followed by the reaction of the alkynide with
To show:
How to synthesize trans- 6-methyl-3 heptene from 4-methyl-1-pentyne.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- The following syntheses have flaws. What is wrong with each?arrow_forwardQ6. Fill in the blanks left in each of the following syntheses: Example: OH H Find the flaw(s): ОН H₂O* H CH3OH. Hº H ОН ОН H CO OCH3 0 MgBr NHANH2 КОН ОН оньсо H₂CO OCH; Harrow_forwardProvide reaction mechanisms for the following transformationsarrow_forward
- 4. Use the reactant below to perform two separate reactions. Give the mechanism(s) and product(s) for each reaction. Show stereochemistry and be clear in your work. Use chair form. KOEt, EtOH t-Bu |||| H3O+arrow_forward6-40 Give the substitution products expected from solvolysis of each compound by heating in ethanol. (a) (b) (c) Br (d) Br CI CH3 Brarrow_forward3. Draw the systematic reaction mechanism for between the reaction of isoamyl alcohol and acetic acid What are the products? What is the functional group of the major product? Does it have a scent? If so, wha is the scent smells like? Choelie on Donio to malke dninl S 00/ ethang1. lohonatou Ilawe:arrow_forward
- 8. Predict the products of the following reactions and propose a complete mechanism for each reaction. 1 Hg(OAc). CH;OH, 2 NABH, CH, CH,CH,CH=CH, 1. BH3. THF 2. H,O». OHarrow_forward2) reaction sequences to carry out those transformations. Draw the expected product of each step proposed. Substantial partial credit can be obtained for incomplete or incorrect answers. You may present your solutions on the following blank page if you need the the following reaction transformations, and propose room. H3C CH, H3C CH3 of HN- -NH HO,C Co,H но H,C CH3 CEN OCH3 OCH3arrow_forwardThe reaction of methylpropene with HBr in ether gives one of the two products below as the major product. Br HBr Br ether Product A Product B Product would have a higher energy transition state for the formation of the intermediate leading to it. O A O B O Both products would have the same transition state.arrow_forward
- Synthesis: Synthesize the following compounds using the starting material shown a reagents you may need. Show the product after each step. You do not need to shc and I will assume standard work up unless you indicate otherwise. HO, ÕHarrow_forwardapp.101edu.co Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side product is continuously removed to drive the reaction toward products. esc F1 CH3CH2CH2OH H2SO4 (cat), heat ☀ F2 80 F3 OH a F4 W Q F5 Atoms and P Draw or ta MacBook A C F6arrow_forwardConsider the reaction scheme below. (i) H3COCHN CO₂Et A H₂N OH EtO OEt B C6H₁1 NO2 base Ha Identify the reagent(s) and conditions A and the intermediate B in this reaction scheme.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

