
Concept explainers
(a)
Interpretation:
The description of the molecule shown, having G as a generic substituent, is to be written.
Concept introduction:
An
Answer to Problem A.1P
The given molecule is a substituted ethane.
Explanation of Solution
The given molecule is:
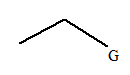
In this molecule, there are two carbon atoms in the straight chain. Hence, the alkane must be ethane
The given molecule is a substituted ethane.
(b)
Interpretation:
The description of the molecule shown, having G as a generic substituent, is to be written.
Concept introduction:
An alkane is said to be substituted if a hydrogen atom of the alkane is replaced by another atom or group of atoms. The atom or group of atoms which replaces the hydrogen atom is called a substituent. It is shown by G, which means a generic substituent.
Answer to Problem A.1P
The given molecule is a substituted pentane.
Explanation of Solution
The given molecule is:
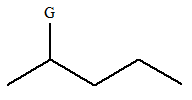
In this molecule, there are five carbon atoms in the straight chain. Hence, the alkane must be pentane
The given molecule is a substituted pentane.
Want to see more full solutions like this?
Chapter A Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Can you help me with the explanation of the alkane, alkene, alkyne, aromatic and all these terms give me examples too?arrow_forwardBased on your answer to below Problem, do you thinkthe compound shown here should have a significantdipole moment? If so, in which direction does it point? The molecule shown here has quite a large dipole, asindicated in its electrostatic potential map. Explain why.Hint: Consider various resonance structures.arrow_forwardAlthough the hypothetical molecule shown here has alternating single and double bonds, those double bonds are not considered to be conjugated. Why not?arrow_forward
- Due to the extra unpair electron in atomic oxygen orbital, it is very much reactive than unsaturated fatty acid. Unsaturated fatty acid contains double bond which is in neutral condition, it is not so much reactive like a double bond please explain this or expoundarrow_forwardPlease help , will provide helpful ratings for solving all 4 subparts only. Draw the skeletal structures that correspond to the following systematic (IUPAC) names.arrow_forwardView the first compound name provided in Table 6. Follow the steps below and draw each part of the structure on a piece of paper. Determine the number of carbons present in the compound based on the base name. Draw the carbon chain and include any double or triple bonds if indicated in the suffix of the base name. Number each carbon. The carbons can be numbered from left to right or right to left. Draw any substituents on the corresponding carbon atom for which is indicated in the name. Refer to Figures 3 and 4 in the background for a visual representation of numbered carbons with corresponding substituents. Check that each carbon atom has a total of 4 bonds.arrow_forward
- In Table what is the smallest alkane, the smallest terminal alkene, and the smallest terminal alkyne that are liquids at room temperature, which is generally taken to be 20 °C to 25 °C?arrow_forwardWhich of the following structural pairs represents contributors to a resonance hybrid? Explain – please show your work for clear understanding.arrow_forwardI know there is KETONE and HYDROXYL group but what else?arrow_forward
- please answer this spec question! answer choices are given except toluene and tetrahydrofuran!arrow_forwardNeed help with question. How are the two products different from each other ?arrow_forwardI don't understand where the H goes for the first product. I'm not sure exactly what the question is asking for. Would this be considered a substitution reaction even though there are two products?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
