
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter E, Problem 3PP
Practice Problem E.3
Kodel is another polyester that enjoys wide commercial use:
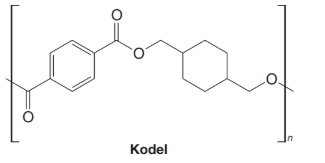
Kodel is also produced by a transesterification. (a) What methyl ester and what alcohol are required for the synthesis of Kodel? (b) The alcohol can be prepared from dimethyl terephthalate. How might this be done?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following diols ARE likely to undergo oxidative change with H2CrO4? Select all that apply
Practice problem 1: Show the correct major organic products. Thank you !
Thapsigargin is a natural product with promising anticancer properties.
Question : At which sites can thapsigargin hydrogen bond to another molecule like itself?
Chapter E Solutions
Organic Chemistry
Ch. E - Prob. 1PPCh. E - Practice Problem E.2
Transesterifications are...Ch. E - Practice Problem E.3
Kodel is another polyester...Ch. E - Practice Problem E.4 Heating phthalic anhydride...Ch. E - Prob. 5PPCh. E - Practice Problem E.6
The familiar “epoxy resins”...Ch. E - Practice Problem E.7 A typical polyurethane can be...Ch. E - Practice Problem E.8
Using a para-substituted...Ch. E - Practice Problem E.9
Outline a general mechanism...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
The difference between an anion and a cation needs to be explained. Concept introduction: The concept related t...
Living by Chemistry
Which of the roll owing compounds have a dipole moment of zero?
Organic Chemistry (8th Edition)
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
Identify the reagents you would use to make each of the following esters:
Organic Chemistry As a Second Language: Second Semester Topics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Hello, I do not understand these questions and I am stuck. May I get help please please?? Question: Synthesize the following compounds using the rules provided using the alkyne shown and other compounds each containing 1 carbon must be incorporated into structure.arrow_forwardOChem Question: Starting with propyne and using any other required reagents, show how you would synthesize (2R, 3R)- and (2S, 3S)-2,3-dibromobutane (as a racemic form)arrow_forwardQuestion: Turn the starting mass of acetanilide to moles, show the calculation with units for full credit. Given starting mass was 1.0 g of acetanilide into a 100 mL beaker. Used 2 g of N bromo succinimde, and 0.230 g of mandelic acid. ( Bromination of acetanilide:electrophilic substitution reaction) Please solve and explain B. Assuming that the acetanilide is the limiting reagent, calculate the theoretical yield of bromo-acetanilide product.arrow_forward
- Organic chemistry question Please provide a well explained solution/answer for the following question. what are electrophilic reagents and please list all possible electrophilic reagents.arrow_forwardProblem B: Dicholorodiflouromethane, CCl2F2, a known refrigerant, can be produced by the chemical equation shown below. Consider the following: atomic mass of C is 12, Cl is 35, and F is 19. CH4 + Cl2 -> CCl4 + HCl (not balanced) CCl4 + HF -> CCl2F2 + HCl (not balanced) 1. Assuming that all the CCl4 produced in the first reaction is consumed in the second, how many moles of Cl2 must be consumed in the first reaction to produce 2.25kg CCl2F2 in the second? Show the complete solution A. 56.24 mol Cl2 B. 56.25 mol Cl2 C. 56.26 mol Cl2 D. 56.27 mol Cl2 E. 2250g Cl2 F. None of the abovearrow_forwardtrue or false question. ( explanation not needed) a) Gilman copper reagent does 1,2 nucleophilic addition to α,β.unsaturated ketones b)Tertiary alcohol will be formed by the reaction between any ketone and Grignard. c)Cyclopentadiene anion is aromatic d)Tertiary alcohols undergoes dehydration by E1 mechanism e)1,4-addition product is the kinetic product in the addition of conjugated diene f) Benzene is more reactive than toluene in aromatic electrophilic substitution reactionarrow_forward
- Hello, May get help with this organic chemistry please? Question: Synthesize the following aromatic from benzenearrow_forwardHello, I do not understand these questions and I am stuck. May I get help please?? Question: draw the MAJOR organic product(s) for the following reactions. If no reaction occurs state no reaction in the box providedarrow_forwardHello, I do not understand these questions and I am stuck. May I get help please please?? Question: Synthesize the following compounds using the rules provided using the alkyne shown and other compounds each containing 1 carbon must be incorporated into structure using the cyclopentene shown and one other 1 carbon compound must be incorporated into final productarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY