3. The following absorbance spectrum was collected. What color is the sample solution? Explain using the color wheel. 1.4 1.2 1. 0.8- 0.6 0.4 0.2+ 300 400 500 600 700 800 wavelength (nm) Absorbance
3. The following absorbance spectrum was collected. What color is the sample solution? Explain using the color wheel. 1.4 1.2 1. 0.8- 0.6 0.4 0.2+ 300 400 500 600 700 800 wavelength (nm) Absorbance
Basic Clinical Lab Competencies for Respiratory Care: An Integrated Approach
5th Edition
ISBN:9781285244662
Author:White
Publisher:White
Chapter14: Introduction To Respiratory Care Pharmacology
Section: Chapter Questions
Problem 5SEPT
Related questions
Question
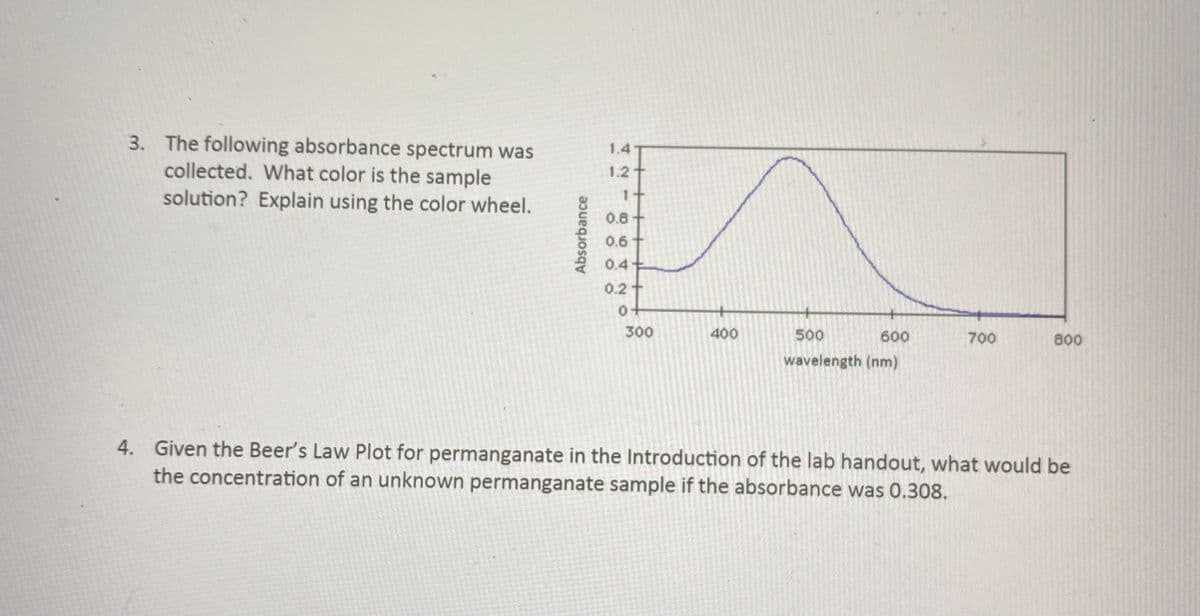
Transcribed Image Text:3. The following absorbance spectrum was
collected. What color is the sample
solution? Explain using the color wheel.
1.4-
1.2
0.8
0.6+
0.4
0.2
300
400
500
600
700
800
wavelength (nm)
4. Given the Beer's Law Plot for permanganate in the Introduction of the lab handout, what would be
the concentration of an unknown permanganate sample if the absorbance was 0.308.
Absorbance
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Basic Clinical Lab Competencies for Respiratory C…
Nursing
ISBN:
9781285244662
Author:
White
Publisher:
Cengage

Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage


Basic Clinical Lab Competencies for Respiratory C…
Nursing
ISBN:
9781285244662
Author:
White
Publisher:
Cengage

Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage



