For the following reaction, polarimetry was used to determine the optical purity of the products. Need help naming two other methods that I could use to determine this and describe how they work. Thank you :
For the following reaction, polarimetry was used to determine the optical purity of the products. Need help naming two other methods that I could use to determine this and describe how they work. Thank you :
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter22: Reactions Of Benzene And Its Derivatives
Section: Chapter Questions
Problem 22.51P
Related questions
Question
For the following reaction, polarimetry was used to determine the optical purity of the products. Need help naming two other methods that I could use to determine this and describe how they work. Thank you :)
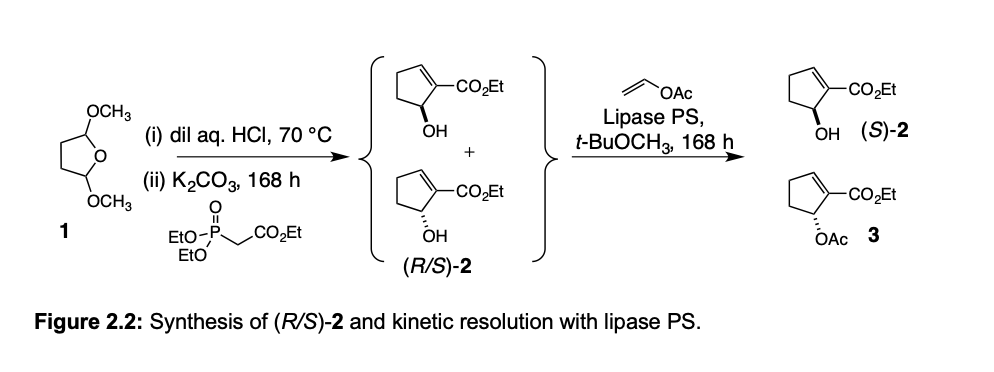
Transcribed Image Text:-CO̟Et
OAc
-co,Et
OCH3
(i) dil aq. HCI, 70 °C
Lipase PS,
t-BUOCH3, 168 h
OH
он (S)-2
(iї) K-СОз, 168 h
OCH3
-CO2Et
1
CO̟Et
Eto-P
EtO
ОН
ОАс 3
(R/S)-2
Figure 2.2: Synthesis of (R/S)-2 and kinetic resolution with lipase PS.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning