KINE DETERMINA RDER OF REACT Table: Volume of KMNO4 Used and Concentration of H2O2 Reaction Time (min) 3 6 9 12 15 18 Volume of KMNO4 (Vina)(mL) 17.5 25.5 31.5 36.0 38.8 42.2 Volume of KMNO4 (Vntia)(mL) 17.5 25.5 31.5 36.0 38.8 Volume of KMNO4 needed (Vinal - Vinitia)(ML) 17.5 8.0 6.0 4.5 2.8 3.4 Concentration of H2O2 (mol dm-3) remained unreacted at time t (using Eq. 1) 0.156 0.136 0.121 0.110 0.103 0.095
KINE DETERMINA RDER OF REACT Table: Volume of KMNO4 Used and Concentration of H2O2 Reaction Time (min) 3 6 9 12 15 18 Volume of KMNO4 (Vina)(mL) 17.5 25.5 31.5 36.0 38.8 42.2 Volume of KMNO4 (Vntia)(mL) 17.5 25.5 31.5 36.0 38.8 Volume of KMNO4 needed (Vinal - Vinitia)(ML) 17.5 8.0 6.0 4.5 2.8 3.4 Concentration of H2O2 (mol dm-3) remained unreacted at time t (using Eq. 1) 0.156 0.136 0.121 0.110 0.103 0.095
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter20: Kinetics
Section: Chapter Questions
Problem 20.89E: Label the elementary processes for the reaction between H2 and O2 see section 20.7 as initiation,...
Related questions
Question
Hi may I ask how to determine the order of reaction from these data? I plotted the graph, but idk what to do after that, because I thought it to be a first order of reaction, but the half time was not constant when I calculated it.
Thank you.
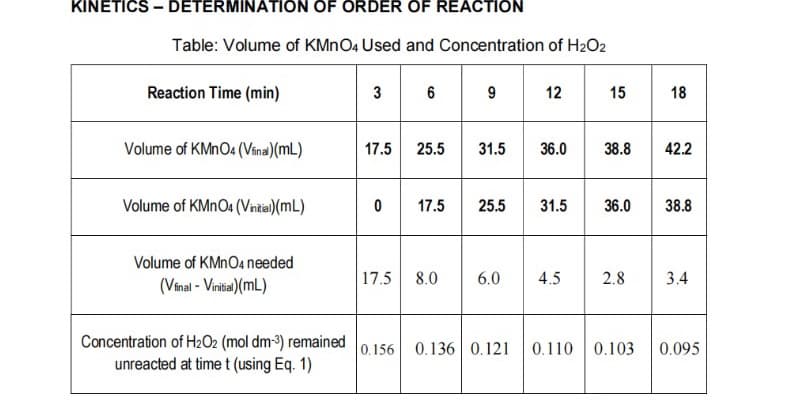
Transcribed Image Text:KINETICS – DETERMINATION OF ORDER OF REACTION
Table: Volume of KMNO4 Used and Concentration of H2O2
3 6 9
• - * *
Reaction Time (min)
12
15
18
Volume of KMNO4 (Vina)(mL)
17.5
25.5
31.5
36.0
38.8
42.2
Volume of KMNO4 (Vntia)(mL)
17.5
25.5
31.5
36.0
38.8
Volume of KMNO4 needed
(Vinal - Vinitia)(mL)
17.5 8.0
6.0
4.5
2.8
3.4
Concentration of H2O2 (mol dm-3) remained
unreacted at time t (using Eq. 1)
0.156
0.136 0.121
0.110 0.103
0.095
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning