Phenol is a stronger acjd than ethanol because. 1.lt is more soluble in water than ethanol so that the phenol molecules have a better chance of donating protons to water molecules. 2. The ethanol molecule is stabilized by delocalization which inhibits the donation of a porton by the OH group. 3.The phenoxide ion is stabilized by delocalization involving the benzene ring, this cannot happen with the ethoxide ion.
Phenol is a stronger acjd than ethanol because. 1.lt is more soluble in water than ethanol so that the phenol molecules have a better chance of donating protons to water molecules. 2. The ethanol molecule is stabilized by delocalization which inhibits the donation of a porton by the OH group. 3.The phenoxide ion is stabilized by delocalization involving the benzene ring, this cannot happen with the ethoxide ion.
Chapter87: Steam Distillation
Section: Chapter Questions
Problem 4P
Related questions
Question
show-all-working-explaining-detailly-each-step.
Answer should be typewritten using a computer keyboard

Transcribed Image Text:Phenol is a stronger acjd than ethanol
because.
41.
1.lt is more soluble in water than ethanol so
that the phenol molecules have a better
chance of donating protons to water
molecules.
2. The ethanol molecule is stabilized by
delocalization which inhibits the donation of
a porton by the OH group.
3.The phenoxide ion is stabilized by
delocalization involving the benzene ring,
this cannot happen with the ethoxide ion.
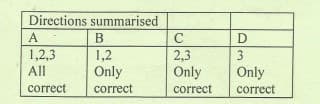
Transcribed Image Text:Directions summarised
A
B
1,2,3
1,2
2,3
Only
All
Only
Only
correct
correct
correct
correct
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax