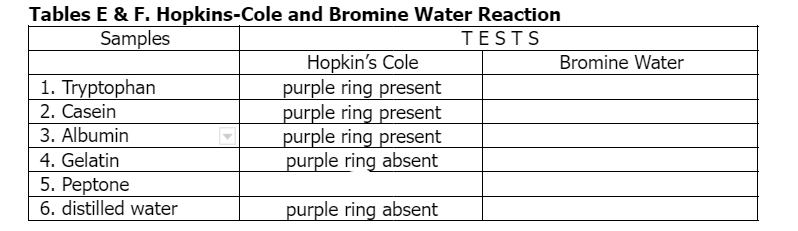
Tables E & F. Hopkins-Cole and Bromine Water Reaction Samples TESTS Hopkin's Cole purple ring present purple ring present purple ring present purple ring absent Bromine Water 1. Tryptophan 2. Casein 3. Albumin 4. Gelatin 5. Peptone 6. distilled water purple ring absent
Tables E & F. Hopkins-Cole and Bromine Water Reaction Samples TESTS Hopkin's Cole purple ring present purple ring present purple ring present purple ring absent Bromine Water 1. Tryptophan 2. Casein 3. Albumin 4. Gelatin 5. Peptone 6. distilled water purple ring absent
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 8.1P
Related questions
Question
color of the bromine water test
Transcribed Image Text:Tables E & F. Hopkins-Cole and Bromine Water Reaction
Samples
TESTS
Hopkin's Cole
purple ring present
purple ring present
purple ring present
purple ring absent
Bromine Water
1. Tryptophan
2. Casein
3. Albumin
4. Gelatin
5. Peptone
6. distilled water
purple ring absent
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage


Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage