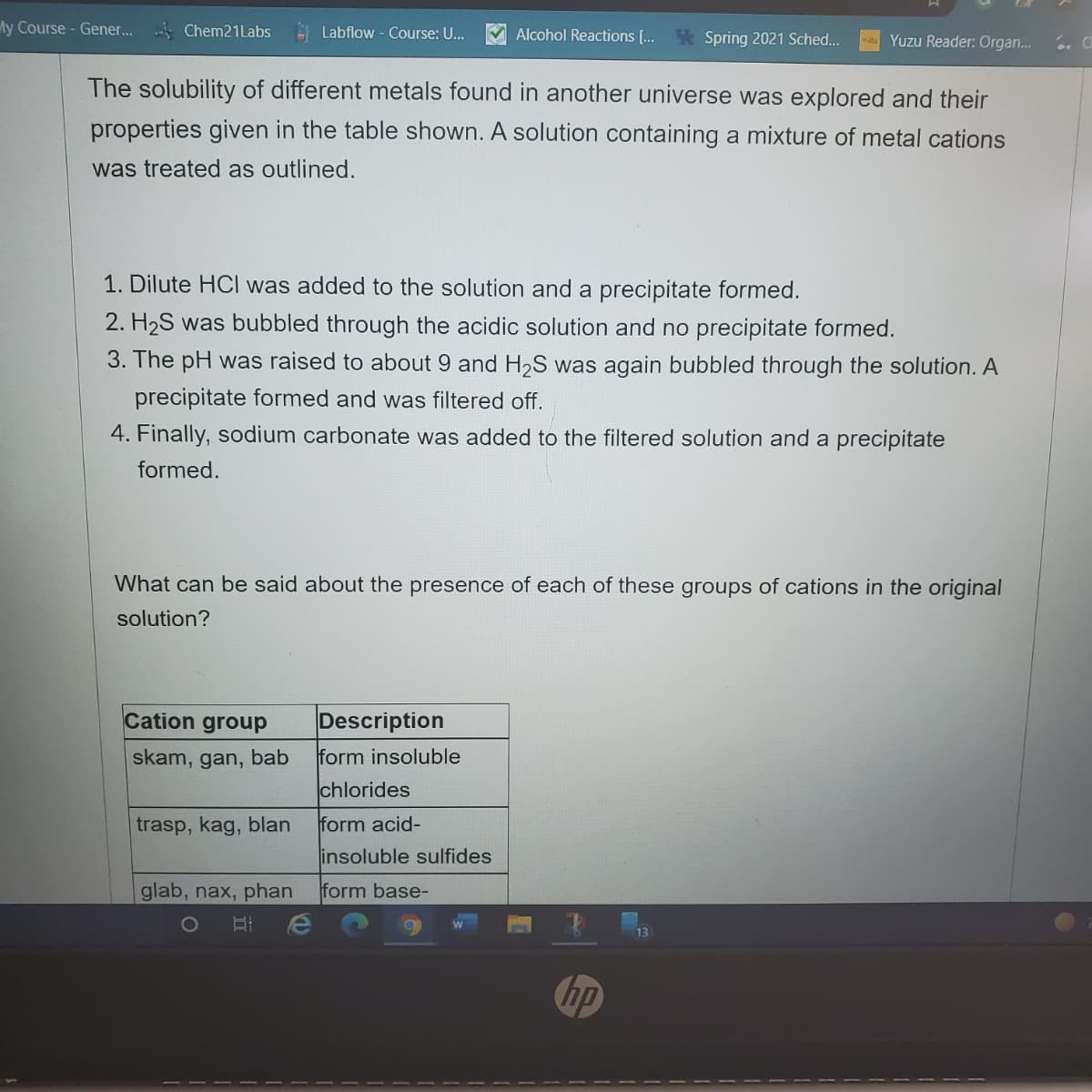
The solubility of different metals found in another universe was explored and their properties given in the table shown. A solution containing a mixture of metal cations was treated as outlined. 1. Dilute HCI was added to the solution and a precipitate formed. 2. H2S was bubbled through the acidic solution and no precipitate formed. 3. The pH was raised to about 9 and H2S was again bubbled through the solution. A precipitate formed and was filtered off. 4. Finally, sodium carbonate was added to the filtered solution and a precipitate formed. What can be said about the presence of each of these groups of cations in the original solution?
The solubility of different metals found in another universe was explored and their properties given in the table shown. A solution containing a mixture of metal cations was treated as outlined. 1. Dilute HCI was added to the solution and a precipitate formed. 2. H2S was bubbled through the acidic solution and no precipitate formed. 3. The pH was raised to about 9 and H2S was again bubbled through the solution. A precipitate formed and was filtered off. 4. Finally, sodium carbonate was added to the filtered solution and a precipitate formed. What can be said about the presence of each of these groups of cations in the original solution?
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 1P
Related questions
Question
Please see attached photo and answer question in photo, for #41. Thanks!
Transcribed Image Text:ly Course - Gener...
Chem21Labs
E Labflow - Course: U...
Alcohol Reactions [..
Spring 2021 Sched...
Yuzu Reader: Organ...
The solubility of different metals found in another universe was explored and their
properties given in the table shown. A solution containing a mixture of metal cations
was treated as outlined.
1. Dilute HCI was added to the solution and a precipitate formed.
2. H2S was bubbled through the acidic solution and no precipitate formed.
3. The pH was raised to about 9 and H2S was again bubbled through the solution. A
precipitate formed and was filtered off.
4. Finally, sodium carbonate was added to the filtered solution and a precipitate
formed.
What can be said about the presence of each of these groups of cations in the original
solution?
Cation group
Description
skam, gan, bab
form insoluble
chlorides
trasp, kag, blan
form acid-
insoluble sulfides
glab, nax, phan
form base-
hp
![Tuzu Rea0
trasp, kag, blan
form acid-
insoluble sulfides
glab, nax, phan form base-
insoluble sulfides
or hydroxides
form insoluble
dapt, zad, pab
carbonates
nant, vant, tant
completely soluble
skam, gan, bab
[ Choose ]
[Choose ]
trasp, kag, blan
At lease one of these ions was present.
All of these ions were present.
None of the ions were present.
glab, nax, phan
Unknown.
dapt, zad, pab
[ Choose ]
nant, vant, tant
[ Choose ]
13
hp](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fd7e141d7-49c5-44f7-9f3c-7e5d88d63be5%2F90b54a71-251e-4b67-85c8-b9f4f1b04576%2Fpaka52n_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Tuzu Rea0
trasp, kag, blan
form acid-
insoluble sulfides
glab, nax, phan form base-
insoluble sulfides
or hydroxides
form insoluble
dapt, zad, pab
carbonates
nant, vant, tant
completely soluble
skam, gan, bab
[ Choose ]
[Choose ]
trasp, kag, blan
At lease one of these ions was present.
All of these ions were present.
None of the ions were present.
glab, nax, phan
Unknown.
dapt, zad, pab
[ Choose ]
nant, vant, tant
[ Choose ]
13
hp
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning