
Concept explainers
As part of a science project, you study traffic patterns in your city at an intersection in the middle of downtown. You set up a device that counts the cars passing through this intersection for a 24-hour period during a weekday. The graph of hourly traffic looks like this.
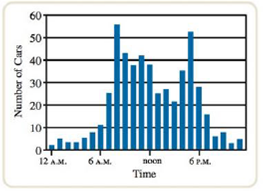
a. At what time(s) does the highest number of cars pass through the intersection?
b. At what time(s) does the lowest number of cars pass through the intersection?
c. Briefly describe the trend in numbers of cars over the course of the day.
d. Provide a hypothesis explaining the trend in numbers of cars over the course of the day.
e. Provide a possible experiment that could test your hypothesis.
Trending nowThis is a popular solution!

Chapter 1 Solutions
Chemistry: Atoms First Approach (Instructor's)
- The mineral fluorite contains the elements calcium and fluorine and can have various colors, including blue, violet, green, and yellow. (a) What are the symbols of these elements? (b) How would you describe the shape of the fluorite crystals in the photo? What can this tell us about the arrangement of the particles (ions) inside the crystal?arrow_forwardCopper: (a) Suppose you have a cube of copper metal that is 0.236 cm on a side with a mass of 0.1206 g. If you know that each copper atom (radius = 128 pm) has a mass of 1.055 1022 g (you will learn in Chapter 2 how to find the mass of one atom), how many atoms are there in this cube? What fraction of the cube is filled with atoms? (Or conversely, how much of the lattice is empty space?) Why is there empty space in the lattice? (b) Now look at the smallest, repeating unit of the crystal lattice of copper. Knowing that an edge of this cube is 361.47 pm and the density of copper is 8.960 g/cm3, calculate the number of copper atoms in this smallest, repeating unit.arrow_forwardAs part of a science project, you study traffic patterns in your city at an intersection in the middle of downtown. You set up a device that counts the ears passing through this intersection for a 24-hr period during a weekday. The graph of hourly traffic looks like this. a. At what time(s) does the highest number of cars pass through die intersection? b. At what time(s) does the lowest number of cars pass through die intersection? c. Briefly describe the trend in numbers of cars over the course of die day. d. Provide a hypothesis explaining the trend in numbers of cars over the course of the day. e. Provide a possible experiment that could test your hypothesis.arrow_forward
- The burning of gasoline in automobile engines is a chemical reaction. In light of the law of conservation of mass, explain what happens to the gasoline in your cars tank as you drive.arrow_forwardChlorine has two prominent isotopes,37Cl and35Cl . Which is more abundant? How do you know?arrow_forwardCalculate the number of atoms in the universe. The following steps will guide you through this calculation: a. Planets constitute less than 1% of the total mass of the universe and can, therefore, be neglected. Stars make up most of the visible mass of the universe, so we need to determine how many atoms are in a star. Stars are primarily composed of hydrogen atoms and our Sun is an average-sized star. Calculate the number of hydrogen atoms in our Sun given that the radius of the Sun is 7108 m and its density is 1.4g/cm3. The volume of a sphere is given by V=(43)r3 (Hint: Use the volume and the density to get the mass of the Sun.) b. The average galaxy (like our own Milky Way galaxy) contains 11011 stars, and the universe contains 1109 galaxies. Calculate the number of atoms in an average galaxy and finally the number of atoms in the entire universe. c. You can hold 11023 atoms in your hand (five copper pennies constitute 1.41023 copper atoms.) How does this number compare with the number of atoms in the universe?arrow_forward
- Sketch the apparatus commonly used for simple distillation in the laboratory, identifying each component.arrow_forwardYou may have noticed that when water boils, you can see bubbles that rise to the surface of the water. Which of the following is inside these bubbles? Explain. a. air b. hydrogen and oxygen gas c. oxygen gas d. water vapor e. carbon dioxide gasarrow_forward3.107 As computer processor speeds increase, it is necessary for engineers to increase the number of circuit elements packed into a given area. Individual circuit elements are often connected using very small copper “wires” deposited directly onto the surface of the chip. In some processors, these copper interconnects are about 22 nm wide. How many copper atoms would be in a 1-mm length of such an interconnect, assuming a square cross section? (The density of copper is 8.96 g/cm3.)arrow_forward
- 1. Does sodium (Na) attract or release electrons? How many electrons are involved in the transfer?2. Does chlorine (Cl) attract or release electrons? How many electrons are involved in the transfer? 3. What do you think would happen if a sodium atom and a chlorine atom bumped into each other? For instance, if you consider the different molecules of gas that exist separately as the air you breathe 4. Welders use helium (He) and argon (Ar) gases to blow over the metal during the welding process. Why would they want to use these gases?5. If 10 atoms of magnesium (Mg) and 10 atoms of chlorine (Cl) could bump into each other, could they combine to form a substance? ______________How many molecules would be made? ________________________Would there be any atoms left over? __________________________Which ones? ______________How many? ________________________6. My friend, the inventor, says that she has just made a translucent, light-weight metal by combining aluminum with…arrow_forward1. Using the measured circumference (c= 21.3 cm) of the glass ornament, calculate the radius in cm. Formula for circumference of a sphere= 2(3.14) r. 2. Using the radius from above, calculate the surface area (cm^2) of the ornament. (Surface area= 4(3.14)r^2) 3. Calculate the mass of the silver that is in the glass ornament. (Final mass-Initial mass) Initial mass- 14.43 Final mass- 14.53 4. The density of silver is 10.5 g/cm^3. What is the volume of the silver metal lining the inside of the glass ornament? 5. Assume that the volume of silver in the ornament can be approximated by the following equation: Volume= Surface area x thickness. Calculate the approximate thickness of the silver lining in cm and convert the answer to picometers (1x10^12 pm= 1m). 6. If the radius of a silver atom is 160 pm, what is the diameter of a silver atom. How many silver atoms thick is the lining?arrow_forwardIn the Millikan oil-drop experiment (see Figure 2.5), the tinyoil drops are observed through the viewing lens as rising,stationary, or falling, as shown here. (a) What causes theirrate of fall to vary from their rate in the absence of an electricfield? (b) Why do some drops move upward? [Section 2.2] The following exercises are divided into sections that dealwith specific topics in the chapter. The exercises are groupedin pairs, with the answers given in the back of the book tothe odd-numbered exercises, as indicated by the red exercisenumbers. Those exercises whose numbers appear in bracketsare more challenging than the nonbracketed exercises.arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





