
Concept explainers
For a particle in a state having the wavefunction
(a)
(c)
(e)
Plot the probabilities versus
(a)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.26E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above expression is simplified as follows.
The probability for the particle having wavefunction
(b)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.26E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above expression is simplified as follows.
The probability for the particle having wavefunction
(c)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.26E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above expression is simplified as follows.
The probability for the particle having wavefunction
(d)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.26E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above expression is simplified as follows.
The probability for the particle having wavefunction
(e)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.26E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above expression is simplified as follows.
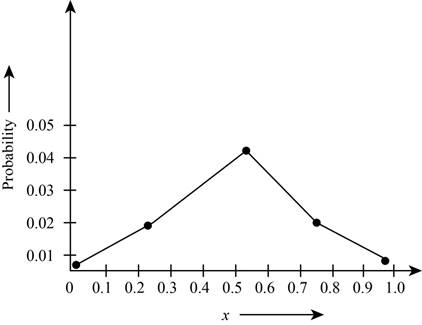
Theplot the probabilities versus

Figure 1
The plot shows the probability for the given wave function. According to this plot, the probability of finding the particle is maximum in the range of
The probability for the particle having wavefunction
Want to see more full solutions like this?
Chapter 10 Solutions
Physical Chemistry
- An electron is accelerated through an electric potential toa kinetic energy of 2.15 x 10-15 J. What is its characteristicwavelength? [Hint: Recall that the kinetic energy of a movingobject is E = 1/2mv2, where m is the mass of the object and n isthe speed of the object.]arrow_forward(a) For a particle in the stationary state n of a one dimensional box of length a, find the probability that the particle is in the region 0xa/4.(b) Calculate this probability for n=1,2, and 3.arrow_forwardA normalized wavefunction for a particle confined between 0 and L in the x direction is ψ = (2/L)1/2 sin(πx/L). Suppose that L = 10.0 nm. Calculate the probability that the particle is (a) between x = 4.95 nm and 5.05 nm, (b) between x = 1.95 nm and 2.05 nm, (c) between x = 9.90 nm and 10.00 nm, (d) between x = 5.00 nm and 10.00 nm.arrow_forward
- For a particle in a box of length L and in the state with n = 3, at what positions is the probability density a maximum? At what positions is the probability density zero?arrow_forwardBy considering the integral ∫02π ψ*ml ψml dϕ, where ml≠m'l, confirm that wavefunctions for a particle in a ring with different values of the quantum number ml are mutually orthogonal.arrow_forwardConsider a collection of 10,000 atoms of rubidium-87, confined inside a box of volume (10-5 m)3. Suppose that T = 0.9Tc. How many atoms are in the ground state? How close is the chemical potential to the ground-state energy? How many atoms are in each of the (threefold-degenerate) first excited states?arrow_forward
- Normalize the wave function ψ= A sin (nπ/a x) by finding the value of the constant A when the particle is restricted to move in one dimensional box of width ‘a’.arrow_forwardA particle freely moving in one dimension x with 0 ≤ x ≤ ∞ is in a state described by the normalized wavefunction ψ(x) = a1/2e–ax/2, where a is a constant. Evaluate the expectation value of the position operator.arrow_forwardConsider a particle of mass m confined to a one-dimensional box of length L and in a state with normalized wavefunction ψn. (a) Without evaluating any integrals, explain why ⟨x⟩ = L/2. (b) Without evaluating any integrals, explain why ⟨px⟩ = 0. (c) Derive an expression for ⟨x2⟩ (the necessary integrals will be found in the Resource section). (d) For a particle in a box the energy is given by En = n2h2/8mL2 and, because the potential energy is zero, all of this energy is kinetic. Use this observation and, without evaluating any integrals, explain why <p2x> = n2h2/4L2.arrow_forward
- For the system described in Exercise E7B.1(a) (A possible wavefunction for an electron in a region of length L (i.e. from x = 0 to x = L) is sin(2πx/L). Normalize this wavefunction (to 1)), what is the probability of finding the electron in the range dx at x = L/2?arrow_forwardWhat values of J may occur in the terms (i) 1S, (ii) 2P, (iii) 3P? How many states (distinguished by the quantum number MJ) belong to each level?arrow_forwardThe moment of inertia of an SF6 molecule is 3.07 × 10−45 kg m2. What is the minimum energy needed to start it rotating?arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
