
Principles of Chemistry: A Molecular Approach (3rd Edition)
3rd Edition
ISBN: 9780321971944
Author: Nivaldo J. Tro
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 64E
The molecule cis-2-butene isomerizes to trans-2-b utene via the following reaction:
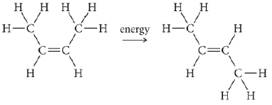
a. If isomerization requires breaking the pi bond, what minimum energy is required for isomerization in J/mol? In J/molecule?
b. If the energy for isomerization comes from light, what minimum
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
5. The reason there is danger in exposure to high-energy radiation (e.g., ultraviolet and X rays)
is that the radiation can rupture chemical bonds. In some cases, cancer can be caused it. A
carbon-carbon single bond has a bond energy of approximately 348 kJ per mole. What
wavelength of light is required to provide sufficient energy to break the C-C bond? In which
region of the electromagnetic spectrum is this wavelength located?
The sun is a giant nuclear reactor, which emits light (electromagnetic radiation) at a range of
frequencies. We human can only see visible light (400 – 700 nm), because its absorption results in a
conformational change in the retinal molecule, which triggers a neural signal.
a.
What is the frequency of a 500 nm visible photon?
b. How much energy in J does a 500 nm visible photon carry?
A typical chemical bond has an energy of 5x10-19 J. Would a visible photon at 500 nm be
С.
sufficient to break a chemical bond?
d. How about a UV (200 nm) photon? Indeed, UV light can damage DNA, the genetic material in
our body, leading to mutations and possibly cancer.
5.a. Explain how the lattice energy of an ionic compound such as KCI can be
determined using the Born-Haber cycle. On what law is this procedure based?
How do the ionization energy and the electron affinity determine whether atoms
of elements will combine to form ionic compounds?
b. Explain in molecular orbital terms the changes in H-H inter-nuclear distance that
occurs as the molecular H2 is ionized first to H2* and then to H2²*. Explain why
the bond order of N2 is greater than that of N2*, but the bond order of 02 is less
than that of O2*.
Chapter 10 Solutions
Principles of Chemistry: A Molecular Approach (3rd Edition)
Ch. 10 - Prob. 10.1PCh. 10 - Prob. 10.2PCh. 10 - Prob. 10.3PCh. 10 -
Predict the geometry of each Interior atom in...Ch. 10 - Prob. 10.5PCh. 10 - Prob. 10.6PCh. 10 - Prob. 10.7PCh. 10 - Prob. 10.8PCh. 10 - Prob. 10.8MPCh. 10 - Prob. 10.9P
Ch. 10 - Prob. 10.10PCh. 10 - Prob. 10.10MPCh. 10 - Prob. 1SAQCh. 10 - Prob. 2SAQCh. 10 - Predict the relative bond angles in BF3 and SO2....Ch. 10 - Prob. 4SAQCh. 10 - Prob. 5SAQCh. 10 - Prob. 6SAQCh. 10 - Prob. 7SAQCh. 10 - Prob. 8SAQCh. 10 - Prob. 9SAQCh. 10 - Prob. 10SAQCh. 10 - Prob. 11SAQCh. 10 - Prob. 12SAQCh. 10 - Prob. 13SAQCh. 10 - Prob. 14SAQCh. 10 - Prob. 15SAQCh. 10 - Prob. 1ECh. 10 - Prob. 2ECh. 10 - Prob. 3ECh. 10 - Prob. 4ECh. 10 - Prob. 5ECh. 10 - Prob. 6ECh. 10 - Prob. 7ECh. 10 - Prob. 8ECh. 10 - Prob. 9ECh. 10 - Prob. 10ECh. 10 - Prob. 11ECh. 10 - Prob. 12ECh. 10 - Prob. 13ECh. 10 - Prob. 14ECh. 10 - Prob. 15ECh. 10 - Prob. 16ECh. 10 - Prob. 17ECh. 10 - Prob. 18ECh. 10 - Prob. 19ECh. 10 - Prob. 20ECh. 10 - Prob. 21ECh. 10 - Prob. 22ECh. 10 - Prob. 23ECh. 10 - Prob. 24ECh. 10 - Prob. 25ECh. 10 - Prob. 26ECh. 10 - Prob. 27ECh. 10 - Prob. 28ECh. 10 - Prob. 29ECh. 10 - Prob. 30ECh. 10 - Prob. 31ECh. 10 - Prob. 32ECh. 10 - 33.Write a hybridization and bonding scheme for...Ch. 10 - Write a hybridization and bonding scheme for each...Ch. 10 - Prob. 35ECh. 10 - Prob. 36ECh. 10 - Prob. 37ECh. 10 - Prob. 38ECh. 10 - Prob. 39ECh. 10 - Prob. 40ECh. 10 - Prob. 41ECh. 10 - Prob. 42ECh. 10 - Prob. 43ECh. 10 - Prob. 44ECh. 10 - Prob. 45ECh. 10 - Prob. 46ECh. 10 - Prob. 47ECh. 10 - Prob. 48ECh. 10 - Prob. 49ECh. 10 - Prob. 50ECh. 10 - Prob. 51ECh. 10 - Prob. 52ECh. 10 - Prob. 53ECh. 10 - The genetic code is based on four different bases...Ch. 10 - Prob. 55ECh. 10 - Prob. 56ECh. 10 - Prob. 57ECh. 10 - Prob. 58ECh. 10 - Prob. 59ECh. 10 - Prob. 60ECh. 10 - Prob. 61ECh. 10 - Prob. 62ECh. 10 - Prob. 63ECh. 10 - The molecule cis-2-butene isomerizes to trans-2-b...Ch. 10 - Prob. 65ECh. 10 - Prob. 66ECh. 10 - Prob. 67ECh. 10 - Prob. 68ECh. 10 - Pick the statement that best captures the...Ch. 10 - Prob. 70ECh. 10 - Prob. 71ECh. 10 - Prob. 72E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A paper published in the research Journal Science in 2007 (S. Vallina and R. Simo, Science, Vol. 315, p. 506, January 26, 2007) reported studies of dimethylsulfide (DMS), an important green-house gas that is released by marine phytoplankton. This gas represents the largest natural source of atmospheric sulfur and a major precursor of hygroscopic (i.e., cloud-forming) particles in clean air over the remote oceans, thereby acting to reduce the amount of solar radiation that crosses the atmosphere and is absorbed by the ocean. (a) Sketch the Lewis structure of dimethylsulfide, CH3SCH3, and list the bond angles in the molecule. (b) Use electronegativities to decide where the positive and negative charges lie in the molecule. Is the molecule polar? (c) The mean seawater concentration of DMS in the ocean in the region between 15 north latitude and 15 south latitude is 2.7 nM (nanomolar). How many molecules of DMS are present in 1.0 m3 of seawater?arrow_forwardAs the bead engineer of your starship in charge of the warp drive, you notice that the supply of dilithium is critically low. While searching for a replacement fuel, you discover some diboron, B2. a. What is the bond order in Li2 and B2? b. How many electrons must be removed from B2 to make it isoelectronic with Li2 so that it might be used in the warp drive? c. The reaction to make B2 isoelectroruc with Li2 is generalized (where n = number of electrons determined in part b) as follows: B2B2n++neE=6455kJ/mol How much energy is needed to ionize 1.5 kg B2 to lhe desired isoelectroruc species?arrow_forwardThe formula for nitryl chloride is CINO2 (in which N is the central atom). a.Draw the Lewis structure for the molecule, including all resonance structures. b.What is the N-O bond order? c.Describe the electron-pair and molecular geometries and give values for all bond angles. d.What is the most polar bond in the molecule? Is the molecule polar? e.The computer program used to calculate electrostatic potential surfaces gave the following charges on atoms in the molecule: A =-0.03, B = -0.26, and C = +0.56. Identify the atoms A, B, and C. Are these calculated charges in accord with your predictions?arrow_forward
- Biotin is being studied in a lab. a. What is the IMF(s) of biotin (C10H16N2O3S)? b. What are the sigma/pi bondings and bond angles of biotin? c. What are the resonance functional groups?arrow_forward32W.) A pi bond is formed by the parallel overlap of s orbitalsX.) Since bromine is from Group VIIA, it needs 7 electrons to complete its octetY.) The bond angle between methane's bond pairs is 109.5⁰Z.) The orbital of f with a shape of y(3x²-y²) contains 14 electronsA.) If all 4 statements are trueB.) If 3 of the 4 statements are trueC.) If 2 of the 4 statements are trueD.) If only 1 of the 4 statements is trueE.) If none of the 4 statements is truearrow_forward4.Examine the model and real structures for SO2. A.In what way does the central atom violate the octet rule? When is it possible for molecules to have this exception to the octet rule? B.Calculate the difference in the bond angle between the real and model structure? Is this difference larger or smaller than the difference observed for H2O? Why is the deviation between the real and model structure different for SO2compared to H2O? The deviation for H2O is 5 degreesarrow_forward
- ||| = 43°F Clear O ELECTRONIC STRUCTURE AND CHEMICAL BONDING Predicting the arrangement of electron groups around the centr... Answer the questions in the table below about the shape of the chlorine pentafluoride (CIF) molecule. How many electron groups are around the central chlorine atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central chlorine atom? (You may need to use the scrollbar to see all the choices.) Explanation Check 10 (choose one) (choose one) linear bent T-shaped trigonal planar trigonal pyramidal square planar square pyramidal tetrahedral sawhorse trigonal bipyramidal octahedral Q Search 0/3 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center www.OTAPETY Accessibiarrow_forwardQuestions 6a, b, c, d is linked to one another and focus on the molecule shown below. H ба) Before we can start assigning hybridization, we need to correctly assign our VSEPR electron-pair geometries to each central atom. For each of the chosen central atom, match it with it's corresponding electron-pair geometry. Drag and drop options on the right-hand side and submit. For keyboard navigation. SHOW MORE V C1 trigonal planar c C2 linear O N1 tetrahedral C trigonal pyramidal N 01 tetrahedral N trigonal planar o Bent O tetrahedral O :0: :0= II II I-0 - -I I-z: Iarrow_forward1. Consider the 1st and 2nd ionization energies of the element Potassium. a. Which of the two ionization energies will be larger? The 1st or 2nd? b. Discuss the number of valence electrons that Potassium has, as well as provide a definition of ionization energy. 2. Consider the carbon dioxide molecule, which is composed of carbon and oxygen. a. Is this molecule polar or non-polar? b. Explain your answer to Part A in terms of molecular geometry of the molecule (draw the Lewis Structure). c. Explain your answer to Part A in terms of electronegativity and the dipole moments found within the carbon dioxide molecule.arrow_forward
- The skeleton structure of atomic chain in 2-aminoacetaldehyde (NH2CH2CHO) is N–C–C–O . The complete Lewis structure of this compound has A. 3 lone pairs, B. 1 π bond and 8 σ bonds. C. 1 lone pair, 1 π bond and 7 σ bonds.D. 3 lone pairs, 1 π bond and 7 σ bonds. E. 1 lone pair, 1 π bond and 8 σ bonds. F 2 lone pairs, 1 π bond and 8 σ bonds.arrow_forwardCompound EX4 reacts with Y2 giving two products: EX3Y and XY. Calculate the E-X bond enthalpy if the reaction liberates 243.6 kJ/mol and other bond enthalpies are: Y-Y 109 kJ/mol, X-Y 123.1 kJ/mol and E-Y 290 kJ/mol. A. 234.7 kJ/mol B. 20.6 kJ/mol C. 547.7 kJ/mol D.176.9 kJ/mol E. 60.5 kJ/molarrow_forwardDraw the Lewis Structure of carbonate ion, CO32-. Please include the resonance structures and the formal charges on each atom. Consider the compound: sulfur tetrafluoride. a. Draw the Lewis Structure, b. What is its AXE notation? c. What is its Molecular geometry? d. What is its Electron pair geometry? e. Is it polar or non-polar? 3. Consider the Ne2 molecule. a. Draw its molecular orbital diagram, b. Compute the bond order, c. Is it more stable compared to its non-bonded form?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY