
(a)
Interpretation:
The species which are used for the transesterification of esters other than acids has to be identified.
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
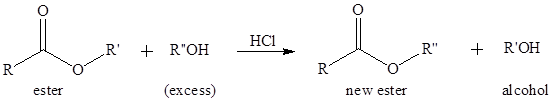
If a better nucleophile is used in place of alcohol molecule that will also increase the rate of transesterification reaction. The conjugate base of an alcohol is an alkoxide which is a better nucleophile and can attack the carbonyl carbon even its oxygen is not protonated.
(b)
Interpretation:
The rate of aminolysis reaction of esters does not increase in presence of
Concept introduction:
The reaction of a carboxylic acid derivative with ammonia, primary and secondary
Trending nowThis is a popular solution!

Chapter 11 Solutions
Essential Organic Chemistry, Global Edition
- Another method to form a peptide bond involves a two-step process:[1] Conversion of a Boc-protected amino acid to a p-nitrophenyl ester.[2] Reaction of the p-nitrophenyl ester with an amino acid ester.a. Why does a p-nitrophenyl ester “activate” the carboxy group of the first amino acid to amide formation? b. Would a p-methoxyphenyl ester perform the same function? Why or why not?arrow_forwardThe reaction of ninhydrin with an -amino acid occurs in several steps (a) The first step is loss of water to give a triketone. Show the mechanism of the reaction and the structure of the triketone.(b) The second step is formation of an imine by reaction of the amino acid with the triketone. Show its structure. (c) The third step is a decarboxylation. Show the structure of the product and the mechanism of the decarboxylation reaction. (d) The fourth step is hydrolysis of an imine to yield an amine and an aldehyde. Show the structures of both products. (e) The final step is formation of the purple anion. Show the mechanism of the reactionarrow_forwardIn the mechanism for cyanohydrin formation, why is HCN the acid that protonates the alkoxide ion instead of HCl?arrow_forward
- The reaction of an ester with an amine is not as slow as the reaction of an ester with water or an alcohol. Explain with reason. Explain why the rate of aminolysis of an ester cannot be increased by H+, OH- or OR-. How can you activate the carboxylic acid? Is acid catalyzed hydrolysis of acetamide a reversible or an irreversible reaction. Explain.arrow_forwarda) Put these three common types of carbonyl compound in order of decreasing reactivity ester amide acid chloride b) For the least reactive, show the interconversion to its other resonance form: How does this electron delocalisation make it stable? c) For the most reactive, draw the mechanism of its undergoing hydrolysis (reaction with H2O): Why makes this type of carbonyl so reactive to nucleophiles?arrow_forwardSynthesis of p-Bromoaniline Why is the protection of the amine function needed in this reaction? a) The protection increases the selectivity for the ortho substitution. b) The protection increases the reactivity of the reactant in the bromination. c) The protection changes the regioselectivity of reaxtion.arrow_forward
- What species other than an acid can be used to increase the rate of the transesterification reaction that converts methyl acetate to propyl acetate?arrow_forwardAccording to the paper, F486 is a phenylalanine residue (i.e., an amino acid that is part of a chain that forms a protein) on SARS-CoV-2. It has an important interaction with ACE2 (best seen in Fig. 1(C) and Fig. 2(A)). Looking at this phenylalanine and the green residues surrounding it, which statement best describes the interaction that could be occurring? a) It forms a hydrogen bond with Y83. b) It forms a dipole-dipole interaction with M82. c) It forms London dispersion forces with surrounding residues. d) It encounters repulsions from surrounding residues.arrow_forwardWhy is an alkylamine more basic than ammononia?arrow_forward
- why I need acetylate amino group and reduce the activity in this mechanism?arrow_forwardHow do you account for the fact that the -COOH group of the conjugate acid of alanine is a stronger acid than the -COOH group of propanoic acid?arrow_forward1. What test reagents are used for the alkylation of amino acids (alkylating reagents)? 2. What test reagents are used for acid hydrolyzation of amino acidsarrow_forward

