
Concept explainers
Interpretation:
In order to generate a carbonyl group in the third product at basic condition, the occurrence has to be discussed.
Concept Introduction:
Stereochemistry: The spatial arrangement of atoms or groups present in compound.
Ring opening: Ring opening of epoxide takes place by acid-catalyzed hydrolysis of an epoxide.
Acid-catalyzed hydrolysis of an epoxide:
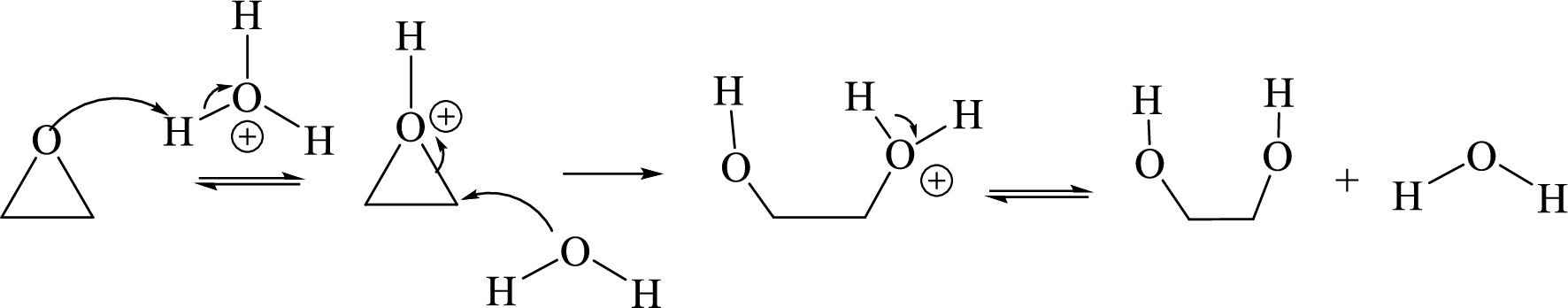
Ease of dehydration of alcohols:
The ease of acid-catalyzed dehydration of alcohols is in the order as follows,
Acid-catalyzed dehydration of primary and secondary alcohols: Dehydration of primary and secondary alcohols is often accompanied by rearrangement process (shift of hydride or alkyl from
Addition of halogen across double bond: The reaction of alkene with halogen molecules undergoes electrophilic addition reaction forming 1,2-dihaloalkane compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry
- draw the major product of this reaction. ignore inorganic byproducts and phthalhydrazide side-product nh2nh2 koharrow_forwardWhat reagents would help me get rid of this Carbonyl oxygenarrow_forwardWhat product is formed when each compound is treated with NaH? Each of these acid–base reactions was a step in a synthesis of a commercially available drug.arrow_forward
- Answer each part for the reaction below. a. Draw in ALL missing lone pairs b. Draw appropriate curved arrows to complete the acid-base reaction c. Draw the corresponding products for the reactionarrow_forwardThe first step in the following reaction is the formation of a(n) _____.arrow_forwardIdentify the reagent necessary to complete this synthesis. Draw the product for each step of synthesis.arrow_forward
- In Chapter 18, we will learn about the hydrolysis of acetals to aldehydes and ketones. Four of the seven steps in the mechanism for this process are shown in the conversion of acetal A to hemiacetal E. a.Add curved arrows for each step. b.Draw another resonance structure for C. c.Identify the nucleophile and electrophile in Step [3]. d.Which steps are Brønsted–Lowry acid–base reactions?arrow_forward14. Adding NaCN to the carbonyl compound poses the risk of making HCN, which is poisonous. How can this risk be mitigated? Add NaOH first, then NaCN Add NaCN first, then NaOH Add NaHSO3 first, then NaCN Add NaOH first, then NaHSO3 NOTE: ONLY ANSWER NUMBER 14arrow_forwardhow to draw in the arrows to show this reaction?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

