
BIOCHEMISTRY-ACHIEVE (1 TERM)
9th Edition
ISBN: 9781319402853
Author: BERG
Publisher: MAC HIGHER
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 12, Problem 4P
Interpretation Introduction
Interpretation:
The diffusion coefficient at 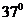 if protein in a membrane of 100 KDa has an effective viscosity of 1 poise and the average distance traveled by 100 kDa protein in
if protein in a membrane of 100 KDa has an effective viscosity of 1 poise and the average distance traveled by 100 kDa protein in 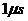
 and
and 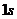 needs to be calculated.
needs to be calculated.
Concept introduction:
Diffusion coefficient also called diffusivity is the diffusing quantity from one region to another passing through unit cross-section area per unit time. It is denoted by D.
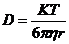
Where,
K =Stefan Boltzmann constant 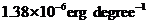
T = Absolute temperature
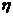 viscosity of the solvent
viscosity of the solvent
r = radius of the sphere.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
More ratios. Through the use of nuclear magnetic resonance
spectroscopy, it is possible to determine the ratio between the
protonated and deprotonated forms of buffers. (a) Suppose the ratio
of [ A- ]A I to [HA] is determined to be 0.1 for a buffer with
pKar6.0.pKa = 6.0. What is the pH? (b) For a different buffer,
91974
suppose the ratio of [ A- ]lA J to [HA] is determined to be 0.1 and
the pHpH is 7.0. In this case, what is the pKapKa of the buffer? (c) For
another buffer with pKa=7.5PKa = 7.5 at pH 8.0pH 8.0, what is the
expected ratio of [ A- ][A ] to [HA]? do
10-¹1 M. A 1 nM (10-⁹ M) solution of lysozyme is
o An Fab fragment binds to lysozyme with a dissociation constant of Kå
treated with increasing concentrations of the Fab fragment. At what concentration of added Fab will half of the lysozyme be
bound to the Fab?
[F] =
nM
Multiple Choice. Imagine a solution containing only water, sodium, and glucose. If a voltage is applied to the solution, glucose would move:
(a) There would be no net movement of glucose
(b) In the opposite direction of sodium
(c) In the same direction and speed as sodium
(d) In the same direction as sodium, but slower.
(e) In the same direction as sodium, but faster.
Chapter 12 Solutions
BIOCHEMISTRY-ACHIEVE (1 TERM)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Sedimenting spheres. What is the dependence of the sedimentation coefficient s of a spherical protein on its mass?How much more rapidly does an 80 -kDa protein sedimentthan does a 40-kDa protein?arrow_forwardCells and Membranes a. Give two examples of molecules that can pass through cell membranes without the need for energy, why can they do so?arrow_forwardChoice and Preparation of a Buffer System1. Choosing the proper buffer solution In Protein Precipitation, two liters of 5mM buffer solution with pH 5.2 is needed in the isolation of albumin. Which among the following buffer solution is best fitted for said purpose? Justify your answer.Buffer solutions pKa Acetate buffer 4.73Tris- (hydroxymethy) aminomethane 8.08Phosphate buffer 7.20 2. Preparation of the chosen buffer system Calculate and measure the amounts (in grams if solid and in mL if liquid) of weak acid and conjugate base needed to be able to prepare the chosen buffer system in part A above. Express your answer in useful units (that is, prepare it from practical amounts or concentrations of starting materials).arrow_forward
- Substituent effects. What is the pHPpH of a 0.1 M0.1 M solution of chloroacetic acid (CICH2 COOH, pKa=2.86)? (CICH, COOH, pK, = 2.86) ? doarrow_forwardTransport. Provided below is an abstracted equation describing an active cell membrane transport from outside the cell to the cytoplasm, involving a membrane carrier (Cmemb), a substrate (Sin) (growth factor etc.) to be carried and released (Sout) into the cytoplasm. a) What kind of feedback mechanism is restricting this process? b) Construct a Forrester Diagram Membrane Transport Carrier, C:C, +S. memb k₁ k_₁ C memb memb + S out Solid Lines. There are two applications in the construction of Forrester diagrams where solid lines can be used to connect state variables. Please name them: a) b)arrow_forwardIsotonicity. Show your complete solution. 1. A hospital pharmacist fills a medication order calling for an intravenous fluid of dextrose 5% in a 0.9% sodium chloride injection and 40 mEq of potassium chloride in a total volume of 1000 mL. The intravenous infusion is administered through an IV set that delivers 15 drops per milliliter. The infusion has been running at a rate of 12 drops per minute for 15 hours. During the 15-hour period: (a) How many mEq of KCl have been administered? (b) How many grams of KCl have been administered? (c) How many millimoles of KCl have been administered? (d) What is the total osmolarity of the intravenous fluid? Express the answer in millimoles (rounded to the nearest whole number) per 1000mL.arrow_forward
- = 20 nM. The rate of receptor-ligand complex formation with an A receptor-ligand complex has a dissociation constant of Ka added ligand concentration of 10 µM is 5 × 10³ s¯¹. What is the value of the reverse rate constant, k_₁ ? k_₁ = 8-1arrow_forward11:14 structure. They provide the matrix or ground substance of extracellular tissue spaces in which collagen and elastin fibers are embedded. Hyaluronic acid, chondroitin 4-sulfate, heparin, are among the important glycosaminoglycans. 10. Glycoproteins are a group of biochemically important compounds with a variable composition of carbohydrate (1-90%), covalently bound to protein. Several enzymes, hormones, structural proteins and cellular receptors are in fact glycoproteins. Chapter 2: CARBOHYDRATES SELF-ASSESSMENT EXERCISES I. Essay questions 1. Define and classify carbohydrates with suitable examples. Add a note on the functions of carbohydrates. 2. Describe the structure and functions of mucopolysaccharides. 3. Give an account of the structural configuration of monosaccharides, with special reference to glucose. 4. Discuss the structure and functions of 3 biochemically important disaccharides. 5. Define polysaccharides and describe the structure of 3 homopolysaccharides. III. Fill…arrow_forwardFrog poison. Batrachotoxin (BTX) is a steroidal alkaloid from the skin of Phyllobates terribilis, a poisonous Colombian frog (the source of the poison used on blowgun darts). In the presence of BTX, Na+Na* channels in an excised patch stay persistently open when the membrane is depolarized. They close when the membrane is repolarized. Which transition is blocked by BTX?arrow_forward
- Hydrolysis .? And what is endocytosis pinocytosis phagocytosis explain with reactions or diagram.arrow_forwardTroubleshooting gels. What effect will be observed on the stained bands of protein in apolyacrylamide gel if…a. …the samples are not incubated at 95˚C prior to loading?b. …the gel is run at too high or too low a voltage?c. …too much protein is loaded?arrow_forwardIonization State of Histidine.Each ionizable group of an amino acid can exist in one of two states, charged or neutral. The electric charge on the functional group is determined by the relationship between its pKa and the pH of the solution. This relationship is described by the Henderson-Hasselbalch equation. 1.Histidine has three ionizable functional groups. Write the equilibrium equations for its three ion-izationsand assign the proper pKa for each ionization. Draw the structure of histidine in each ionization state.What is the net charge on the histidine molecule in each ionization state? 2.Which structure drawn in (1) corresponds to theionization state of histidine at pH 1, 4, 8, and12?Note that the ionization state can be approximated by treating each ionizable group independently. 3.What is the net charge of histidine at pH 1, 4, 8, and 12? For each pH, will histidine migrate to-ward the anode (+) or cathode (-) when placed in an electric field?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY