
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.SE, Problem 48AP
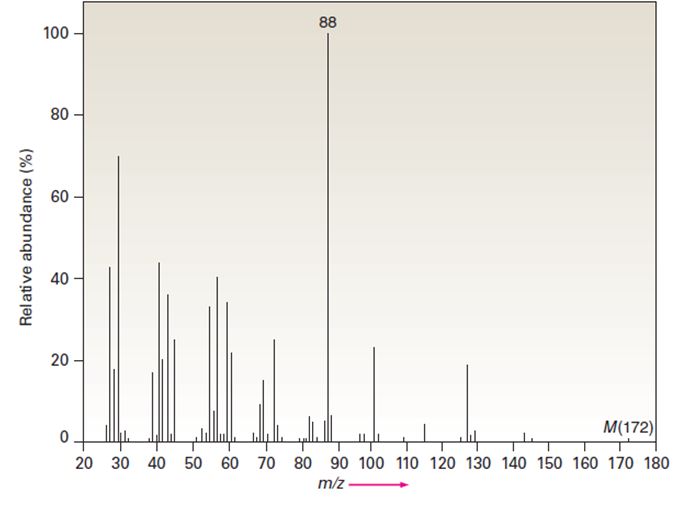
The infrared spectrum of the compound with the mass spectrum shown below lacks any significant absorption above 3000 cm-1. There is a prominent peak near 1740 cm-1 and another strong peak near 1200 cm-1. Propose a structure consistent with the data.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Compound E is a volatile organic liquid with a fruity smell. Its mass spectrum is given below.
Identify the fragments at m/z 77 and 105, then propose a structure for this compound.
Propose a structure consistent with each set of data.
C10H14: IR absorptions at 3150–2850, 1600, and 1500 cm−1
Predict the structure of the compound using the information below. Kindly show what information are obtained for each spectra which may help in predicting the structure.
From IR Spectra:
See the attached picture
From Mass Spectra
M+ = 134
base peak = 105
peaks at 77 and 51
From Proton and Carbon NMR Spectra
See attached picture.
Chapter 12 Solutions
Organic Chemistry
Ch. 12.2 - Prob. 1PCh. 12.2 - Two mass spectra are shown in FIGURE 12-8. One...Ch. 12.3 - What are the masses of the charged fragments...Ch. 12.3 - Prob. 4PCh. 12.5 - Prob. 5PCh. 12.5 - Prob. 6PCh. 12.7 - What functional groups might the following...Ch. 12.7 - How might you use IR spectroscopy to distinguish...Ch. 12.8 - Prob. 9PCh. 12.8 - Where might the following compounds have IR...
Ch. 12.8 - Where might the following compound have IR...Ch. 12.SE - Prob. 12VCCh. 12.SE - Show the structures of the fragments you would...Ch. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - Write molecular formulas for compounds that show...Ch. 12.SE - Camphor, a saturated monoketone from the Asian...Ch. 12.SE - The nitrogen rule of mass spectrometry says that a...Ch. 12.SE - In light of the nitrogen rule mentioned in Problem...Ch. 12.SE - Nicotine is a diamino compound isolated from dried...Ch. 12.SE - The hormone cortisone contains C, H, and O, and...Ch. 12.SE - Halogenated compounds are particularly easy to...Ch. 12.SE - Prob. 22APCh. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - 2-Methylpentane (C6H14) has the mass spectrum...Ch. 12.SE - Assume that you are in a laboratory carrying out...Ch. 12.SE - What fragments might you expect in the mass...Ch. 12.SE - How might you use IR spectroscopy to distinguish...Ch. 12.SE - Would you expect two enantiomers such as...Ch. 12.SE - Would you expect two diastereomers such as meso-2,...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - How could you use infrared spectroscopy to...Ch. 12.SE - Prob. 32APCh. 12.SE - At what approximate positions might the following...Ch. 12.SE - How would you use infrared spectroscopy to...Ch. 12.SE - At what approximate positions might the following...Ch. 12.SE - Assume that you are carrying out the dehydration...Ch. 12.SE - Assume that you are carrying out the base-induced...Ch. 12.SE - Prob. 38APCh. 12.SE - Carvone is an unsaturated ketone responsible for...Ch. 12.SE - Prob. 40APCh. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - 4-Methyl-2-pentanone and 3-methylpentanal are...Ch. 12.SE - Grignard reagents undergo a general and very...Ch. 12.SE - Ketones undergo a reduction when treated with...Ch. 12.SE - Nitriles, R–=C≡N, undergo a hydrolysis...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - Prob. 50AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The infrared spectrum of the compound with the mass spectrum shown below has a medium-intensity peak at about 1650 cm-1. There is also a C-H out-of-plane bending peak near 880 cm-1. Propose a structure consistent with the data.arrow_forwardGive the structure for two isomers of molecular formula C4H10O which are consistent with the ^1H-NMR spectra shown below.arrow_forwardcompound with the molecular formula C7H9N exhibits IR bands at 3450 cm-1 (medium, doublet), and 855 cm-1 (strong) and shows the following major mass spectral signals (m/z): 106 (base); 107 (M+, about 70% of base), 91 (40% of base), and 77 (about 20% of base). Deduce a reasonable structure from this data.arrow_forward
- The infrared spectrum of the compound with the mass spectrum shown below lacks any significant absorption above 3000 cm-1. There is a distinct peak near 1740 cm-1 and another strong peak near 1200 cm-1. molecular ion: M+ = 172arrow_forwardPropose a structure consistent with following set of spectral data: C3H6Br2: IR peak at 3000–2850 cm−1; NMR (ppm): 2.4 (quintet) 3.5 (triplet)arrow_forwardAn compound that has a molecular formula of C5H11NO, has the below 1H NMR spectrum which is showen in the picture. There is a strong single peaked absorption at 3400 and 1710 cm-1and there are couple of mdium peaks between 2800 and 3000 cm-1. Which of the options below is more consistent with this spectrum?arrow_forward
- Propose a structure consistent with each set of spectral data: a. C4H8Br2: IR peak at 3000–2850 cm−1; NMR (ppm): 1.87 (singlet, 6 H) 3.86 (singlet, 2 H) b.C3H6Br2: IR peak at 3000–2850 cm−1; NMR (ppm): 2.4 (quintet) 3.5 (triplet) c. C5H10O2: IR peak at 1740 cm−1; NMR (ppm): 1.15 (triplet, 3 H) 2.30 (quartet, 2 H) 1.25 (triplet, 3 H) 4.72 (quartet, 2 H) d.C3H6O: IR peak at 1730 cm−1; NMR (ppm): 1.11 (triplet) 2.46 (multiplet) 9.79 (triplet)arrow_forwardPropose the structure of the following compound (C) based on its spectral data. Mass spectroscopy gives a predicted molecular weight of 136 and a Formula of C9H12O. No UV was taken. (Image of compound C is attached)arrow_forwardIn the presence of a small amount of acid, a solution of acetaldehyde (CH3CHO) in methanol (CH3OH) was allowed to stand and a new compound L was formed. L has a molecular ion in its mass spectrum at 90 and IR absorptions at 2992 and 2941 cm−1. L shows three signals in its 13C NMR at 19, 52, and 101 ppm. The 1H NMR spectrum of L is given below. What is the structure of L?arrow_forward
- Please show complete answer. Propose a structure consistent with following set of spectral data: a. C5H10O2: IR peak at 1740 cm−1; NMR (ppm): 1.15 (triplet, 3 H) 2.30 (quartet, 2 H) 1.25 (triplet, 3 H) 4.72 (quartet, 2 H) b. C3H6O: IR peak at 1730 cm−1; NMR (ppm): 1.11 (triplet) 2.46 (multiplet) 9.79 (triplet)arrow_forwardReaction of pentanoyl chloride (CH3CH2CH2CH2COCl) with lithium dimethyl cuprate [LiCu(CH3)2] forms a compound J that has a molecular ion in its mass spectrum at 100, as well as fragments at m/z = 85, 57, and 43 (base). The IR spectrum of J has strong peaks at 2962 and 1718 cm-1. Propose a structure for J.arrow_forwardPART 2: Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given below. Information from the DEPT 13C NMR spectra is given near each peak. Give structure for C.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY