
ORGANIC CHEMISTRY
5th Edition
ISBN: 9781259977596
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.20P
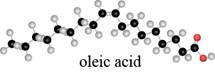
What are the major IR absorptions in the functional group region for oleic acid, a common unsaturated fatty acid (Section 10.6A)?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can the peaks for C–H bonds in alkanes and alkenes be differentiated spectroscopically? If so, briefly explain how. If not, explain why not?
a) which bond is present in alcohols but not alkanes?
b) is this band strong or weak (in terms of spectra)?
c) what is different about C=O and O-H bonds compared with C=C or C≡C bonds that show weaker signals?
10. Provide a structure of an isomer that has the molecular formula C5H8O2 and fits the criteria described below.
Note that all of the structures must be neutral and not have any formal charges on any of the atoms.
a) Contains an ester and an alkene
b) A chiral compound that
contains a ring, ether and ketone.
c) Contains an alcohol and an alkyne.
d) A meso compound that contains two alcohols.
Chapter 13 Solutions
ORGANIC CHEMISTRY
Ch. 13 - What is the mass of the molecular ion formed from...Ch. 13 - Prob. 13.2PCh. 13 - Use the following information to propose a...Ch. 13 - Prob. 13.4PCh. 13 - What molecular ions would you expect for the...Ch. 13 - The mass spectrum of 2,3-dimethylpentane also...Ch. 13 - The base peak in the mass spectrum of 2, 2,...Ch. 13 - (a) What mass spectral fragments are formed by ...Ch. 13 - What cations are formed in the mass spectrometer...Ch. 13 - The low-resolution mass spectrum of an unknown...
Ch. 13 - Benzene, toluene, and p-xylene BTX are often added...Ch. 13 - Prob. 13.12PCh. 13 - Prob. 13.13PCh. 13 - Prob. 13.14PCh. 13 - Prob. 13.15PCh. 13 - How do the IR spectra of the isomers cyclopentane...Ch. 13 - Problem 13.17 How do the three isomers of...Ch. 13 - Problem 13.18 What functional groups are...Ch. 13 - Problem-13.19 What are the major IR absorptions in...Ch. 13 - Problem-13.20 What are the major IR absorptions in...Ch. 13 - Problem-13.21 Which of the following possible...Ch. 13 - Problem-13.22 Propose structures consistent with...Ch. 13 - 13.23 What major IR absorptions are present above ...Ch. 13 - Problem-13.24 The mass spectrum of the following...Ch. 13 - Prob. 13.25PCh. 13 - Which compound gives a molecular ion at m/z= 122,...Ch. 13 - Propose two molecular formulas for each molecular...Ch. 13 - Propose four possible structures for a hydrocarbon...Ch. 13 - Problem-13.29 What is the molecular formula for...Ch. 13 - Problem-13.30 Propose a molecular formula for rose...Ch. 13 - 13.31 Match each structure to its mass spectrum
Ch. 13 - 13.32 Propose two possible structures for a...Ch. 13 - 13.33 What cations are formed in the mass...Ch. 13 - 13.34 and have the same molecular ion in the...Ch. 13 - 13.35 For each compound, assign likely...Ch. 13 - Prob. 13.36PCh. 13 - 13.37 Propose a structure consistent with each...Ch. 13 - 13.38 A low-resolution mass spectrum of the...Ch. 13 - 13.39 Primary alcohols often show a peak in their...Ch. 13 - 13.40 Like alcohols, ethers undergo α cleavage by...Ch. 13 - 13.41 Which of the highlighted bonds absorbs at...Ch. 13 - 13.42 What major IR absorptions are present above ...Ch. 13 - 13.43 How would each of the following pairs of...Ch. 13 - 13.44 Morphine, heroin, and oxycodone are three...Ch. 13 - 13.45 Reduction of cyclohex-2-enone can yield...Ch. 13 - Prob. 13.46PCh. 13 - 13.47 Match each compound to its IR spectrum
Ch. 13 - 13.48 Propose possible structures consistent with...Ch. 13 - A chiral hydrocarbon X exhibits a molecular ion at...Ch. 13 - 13.50 A chiral compound has a strong absorption...Ch. 13 - 13.51 Treatment of benzoic acid with followed by...Ch. 13 - 13.52 Treatment of benzaldehyde with in aqueous ...Ch. 13 - Prob. 13.53PCh. 13 - 13.54 Reaction of 2-methylpropanoic acid with ...Ch. 13 - 13.55 Reaction of pentanoyl chloride with lithium...Ch. 13 - Prob. 13.56PCh. 13 - 13.57 Treatment of anisole with and forms P,...Ch. 13 - 13.58 Reaction of with forms compound ,...Ch. 13 - Problem-13.59 The carbonyl absorption of an amide...Ch. 13 - Prob. 13.60PCh. 13 - Problem-13.61 Explain why a ketone carbonyl...Ch. 13 - 13.62 Oxidation of citronellol, a constituent of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
Which of the following solutions has the higher molarity? 10 ppm KI in water or 10,000 ppb KBr in water 0.25 ma...
CHEMISTRY-TEXT
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- DHA is a fatty acid derived from fish oil and an abundant fatty acid in vertebrate brains. Hydrogenation of DHA forms docosanoic acid [CH3(CH2)20CO2H], and ozonolysis forms CH3CH2CHO, CH2(CHO)2 (five equivalents), and HCOCH2CH2CO2H. What is the structure of DHA if all double bonds have the Z configuration?arrow_forwardUse the IR to determine which functional group is present in the molecule w Alcohol Aldehyde OKetone O Aromatic ring m MENINGarrow_forwardparce Nitroethylene, 1,C-CHNO, is a sensitive compound that must be prepared with great care. Attempted purification of nitroethyfene by distillation often results in low ecovery of product and a white coating on the inner walls of the distillation apparatus. Draw a structure for the white coating. • You do not have to consider stereochemestry • You do not have to explcitly draw H atoms. • Do not incude lone pairs in your answer. They will not be considered in the grading • In cases where there is more than one answer, just draw one. • Use RI groups to indicate the points where the polymer repeats. The R group tool is located in the charges and lone pain op down menu ChanCoodarrow_forward
- The infrared spectrum of a compound is unique to the types and number of bonds in the molecule. Draw the structures of salicylic acid and aspirin. What are the key differences of the IR spectrum of aspirin with that of the starting material, salicylic acid? What are the similarities? Interpret the IR spectra of each by assigning the major peaks.arrow_forwardFor each pair of compounds, predict which one has the higher molecular dipole moment, and explain your reasoning.(a) 1-bromopropane or cyclopropanearrow_forwardMethionine is an amino acid used in the biosynthesis of proteins. The structural diagram for methionine is: H H H H H H H °N H H Using VSEPR theory, consider the stereochemical diagram that would form. Identify t geometric shape at the six identified locations on the above molecule of methionine. Hint: Treat each location as a separate central atom. Remember to add in lone pairs Review this example from your course. (Unit A Section 3 Lesson 8.2 - Digging Deeper) Geometric shape around atom 1 is tetrahedral Geometric shape around atom 2 is tetrahedral Geometric shape around atom 3 is tetrahedral Geometric shape around atom 4 is trigonal planar Geometric shape around atom 5 is tetrahedral Geometric shape around atom 6 is trigonal pyramidal +arrow_forward
- What are the rules surrounding how to determine whether a nitrogen is a stereocenter? Do lone pairs and hydrogens count as a fourth group? What really makes nitrogen a stereocenter?arrow_forwardStereoisomers share the same connectivity and differ only in the way their atoms are arranged in space. Draw the structure of a compound that is a stereoisomer ofarrow_forwardThe naphthalene molecule has a structure that corre- sponds to two benzene molecules fused together: The ™ electrons in this molecule are delocalized over the entire molecule. The wavelength of maximum absorption in benzene is 255 nm. Will the corresponding wavelength in naphthalene be shorter or longer than 255 nm?arrow_forward
- For which compound containing a heteroatom (an atom other than carbon or hydrogen) does the molecular ion have an even-numbered mass? For which does it have an odd-numbered mass? Q.) A bromoalkane with the molecular formula CnH2n11Brarrow_forwardFollowing is a structural formula for cortisol (hydrocortisone). Draw a stereo-representation of this molecule showing the conformations of the five- and six-membered rings.arrow_forwardIt is easy to imagine a cyclohexane as a flat hexagon and a lot of the time we draw it that way. Looking at 1,3,5-triethylcyclohexane we cannot tell the stability of the molecule from looking at the flat 2D drawing. Explain why we need to look at the 3D configuration and what conformation (axial,equatorial) would each of the three ethyl groups be in for the most stable configuration.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY